QUESTION IMAGE
Question
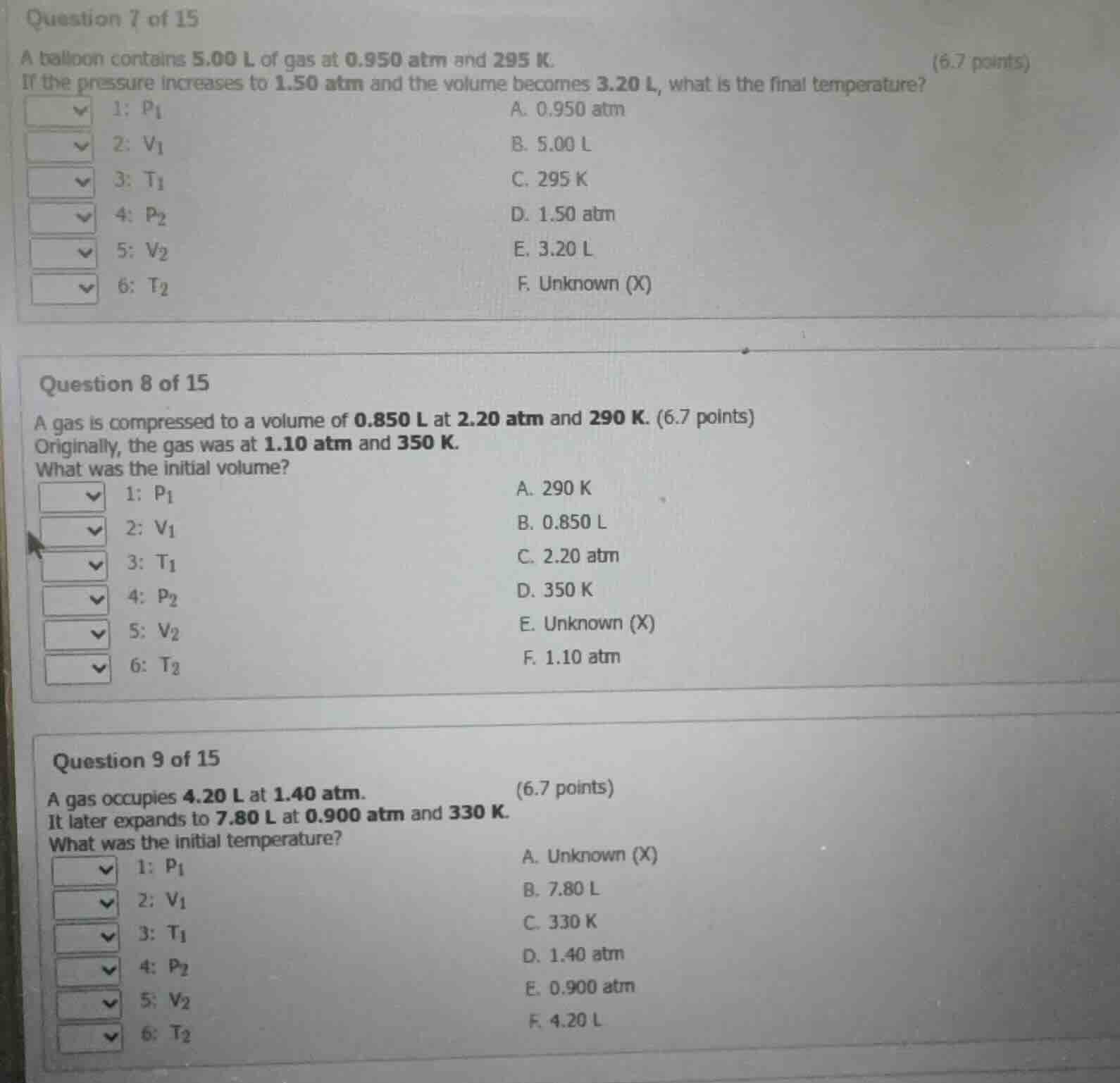
question 7 of 15
a balloon contains 5.00 l of gas at 0.950 atm and 295 k.
if the pressure increases to 1.50 atm and the volume becomes 3.20 l, what is the final temperature?
1: ( p_1 ) a. 0.950 atm
2: ( v_1 ) b. 5.00 l
3: ( t_1 ) c. 295 k
4: ( p_2 ) d. 1.50 atm
5: ( v_2 ) e. 3.20 l
6: ( t_2 ) f. unknown (x)
question 8 of 15
a gas is compressed to a volume of 0.850 l at 2.20 atm and 290 k.
originally, the gas was at 1.10 atm and 350 k.
what was the initial volume?
1: ( p_1 ) a. 290 k
2: ( v_1 ) b. 0.850 l
3: ( t_1 ) c. 2.20 atm
4: ( p_2 ) d. 350 k
5: ( v_2 ) e. unknown (x)
6: ( t_2 ) f. 1.10 atm
question 9 of 15
a gas occupies 4.20 l at 1.40 atm.
it later expands to 7.80 l at 0.900 atm and 330 k.
what was the initial temperature?
1: ( p_1 ) a. unknown (x)
2: ( v_1 ) b. 7.80 l
3: ( t_1 ) c. 330 k
4: ( p_2 ) d. 1.40 atm
5: ( v_2 ) e. 0.900 atm
6: ( t_2 ) f. 4.20 l
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1: A. 0.950 atm
2: B. 5.00 L
3: C. 295 K
4: D. 1.50 atm
5: E. 3.20 L
6: F. Unknown (X)
1: F. 1.10 atm
2: E. Unknown (X)
3: D. 350 K
4: C. 2.20 atm
5: B. 0.850 L
6: A. 290 K
1: D. 1.40 atm
2: F. 4.20 L
3: A. Unknown (X)
4: E. 0.900 atm
5: B. 7.80 L
6: C. 330 K