QUESTION IMAGE
Question
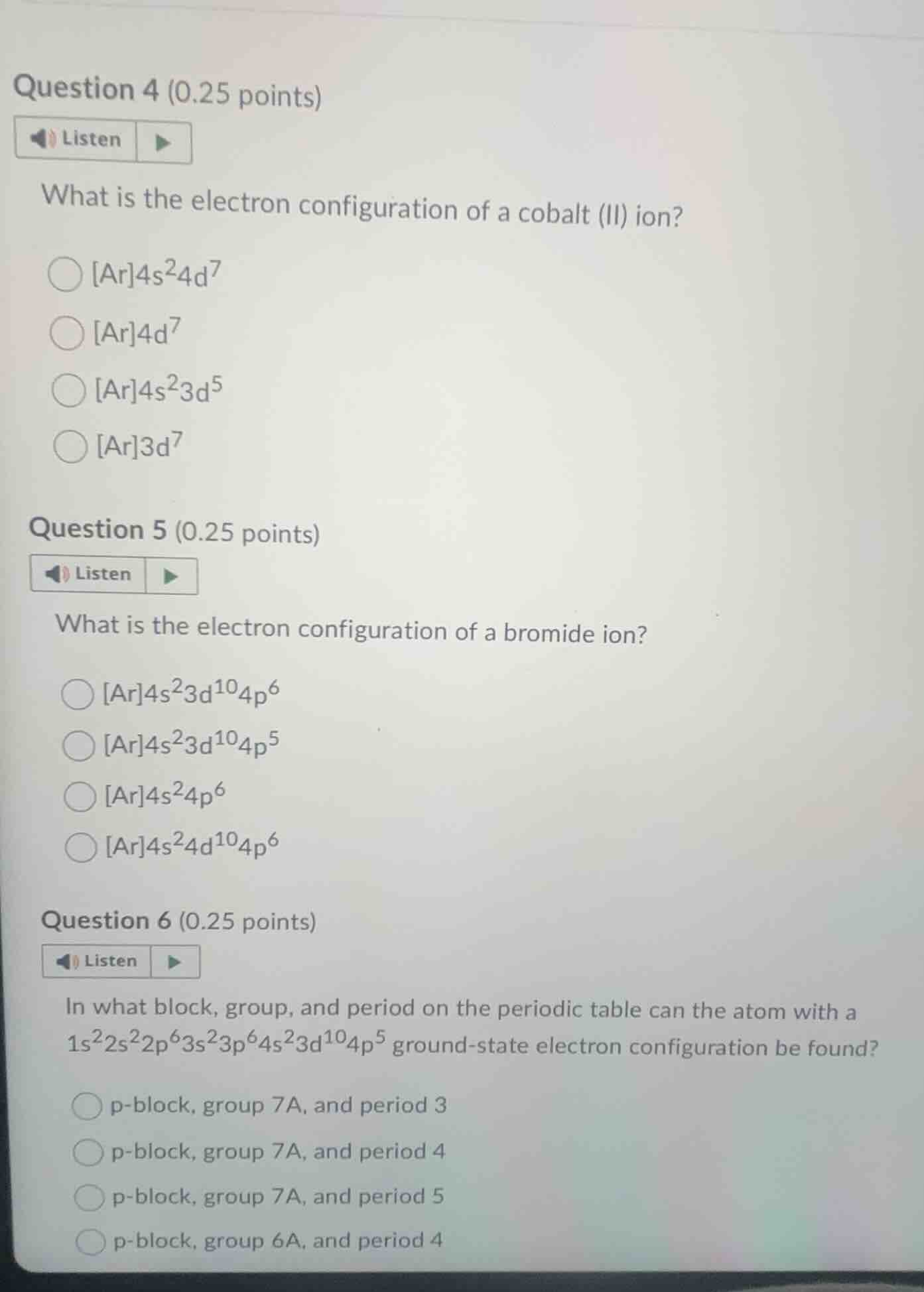
question 4 (0.25 points)listenwhat is the electron configuration of a cobalt (ii) ion?$ar4s^{2}4d^{7}�LXB0�ar4s^{2}3d^{5}�LXB1�ar4s^{2}3d^{10}4p^{5}�LXB2�ar4s^{2}4d^{10}4p^{6}$question 6 (0.25 points)listenin what block, group, and period on the periodic table can the atom with a $1s^{2}2s^{2}2p^{6}3s^{2}3p^{6}4s^{2}3d^{10}4p^{5}$ ground-state electron configuration be found?p-block, group 7a, and period 3p-block, group 7a, and period 4p-block, group 7a, and period 5p-block, group 6a, and period 4
Question 4
Step1: Find neutral Co electron count
Cobalt (Co) has an atomic number of 27, so neutral Co has 27 electrons. Its electron configuration is $[Ar]4s^23d^7$.
Step2: Remove 2 electrons for Co(II)
For transition metals, 4s electrons are lost first. Remove 2 electrons from the 4s orbital: $[Ar]3d^7$.
Question 5
Step1: Find neutral Br electron count
Bromine (Br) has an atomic number of 35, so neutral Br has 35 electrons. Its electron configuration is $[Ar]4s^23d^{10}4p^5$.
Step2: Add 1 electron for Br⁻
A bromide ion ($Br^-$) gains 1 electron, filling the 4p orbital: $[Ar]4s^23d^{10}4p^6$.
Question 6
Step1: Identify highest energy level
The outermost principal energy level is $n=4$, so the period is 4.
Step2: Identify valence electrons
Valence electrons are $4s^24p^5$, total 7 valence electrons, so group 7A. The last electron fills a p orbital, so it is in the p-block.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 4: [Ar]3d⁷
Question 5: [Ar]4s²3d¹⁰4p⁶
Question 6: p-block, group 7A, and period 4