QUESTION IMAGE
Question
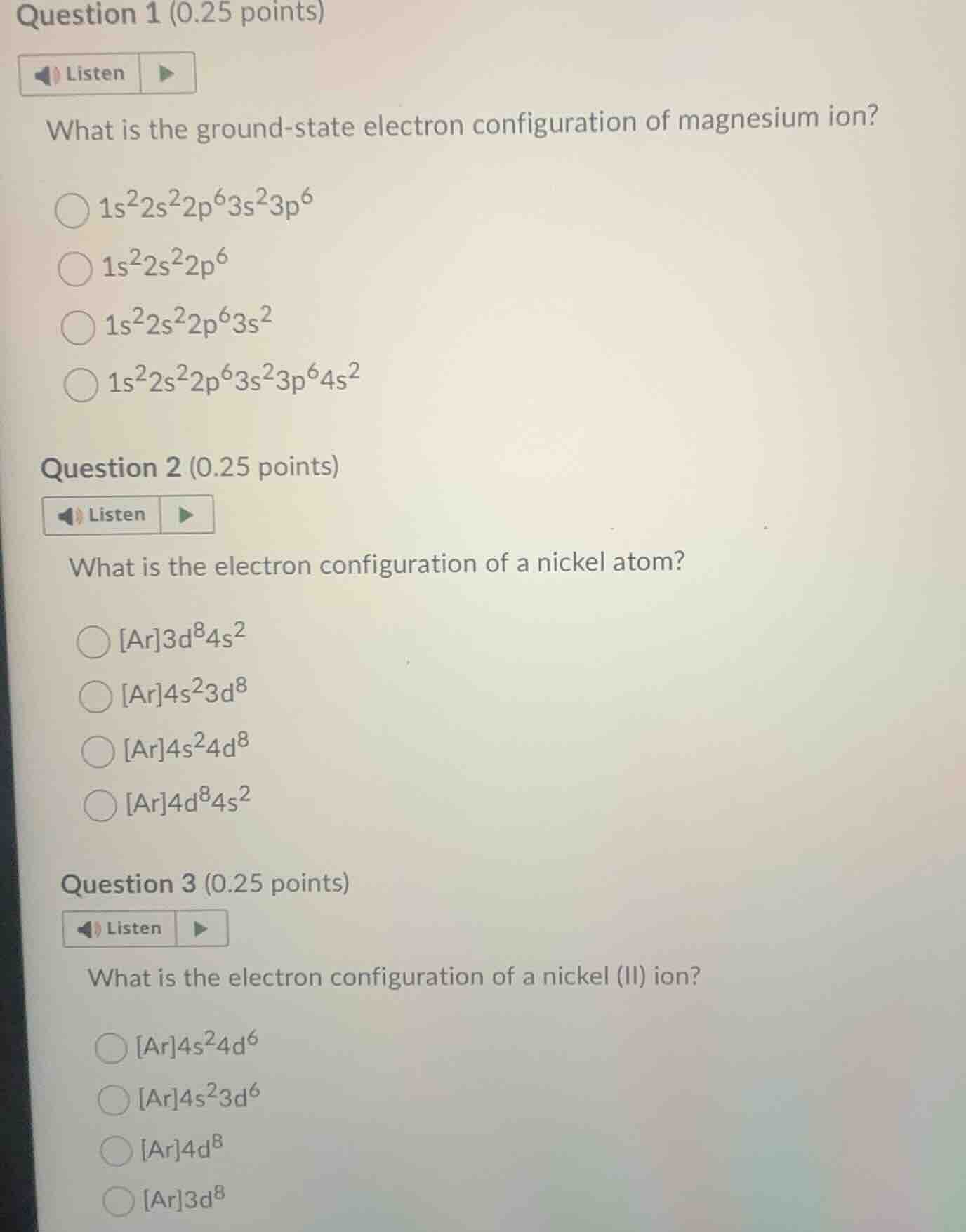
question 1 (0.25 points)listenwhat is the ground-state electron configuration of magnesium ion?$1s^22s^22p^63s^23p^6�LXB0�1s^22s^22p^63s^2�LXB1�ar4s^23d^8�LXB2�ar4d^84s^2$question 3 (0.25 points)listenwhat is the electron configuration of a nickel (ii) ion?$ar4s^24d^6$$ar4s^23d^6$$ar4d^8$$ar3d^8$
Step1: Find Mg atom electron count
Magnesium (Mg) has an atomic number of 12, so a neutral Mg atom has 12 electrons. Its ground-state configuration is $1s^22s^22p^63s^2$.
Step2: Derive Mg²⁺ ion configuration
Magnesium forms a +2 ion by losing 2 electrons from the outermost 3s orbital. Subtract 2 electrons: $1s^22s^22p^6$.
Step3: Find neutral Ni electron count
Nickel (Ni) has an atomic number of 28. Argon (Ar, Z=18) is the preceding noble gas. The remaining 10 electrons fill $4s^2$ first, then $3d^8$, so configuration is $[Ar]4s^23d^8$.
Step4: Derive Ni(II) ion configuration
Ni(II) is a +2 ion, losing 2 electrons from the higher-energy 4s orbital first. Remove 2 electrons from $4s^2$: $[Ar]3d^8$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 1: $1s^22s^22p^6$
Question 2: $[Ar]4s^23d^8$
Question 3: $[Ar]3d^8$