QUESTION IMAGE
Question
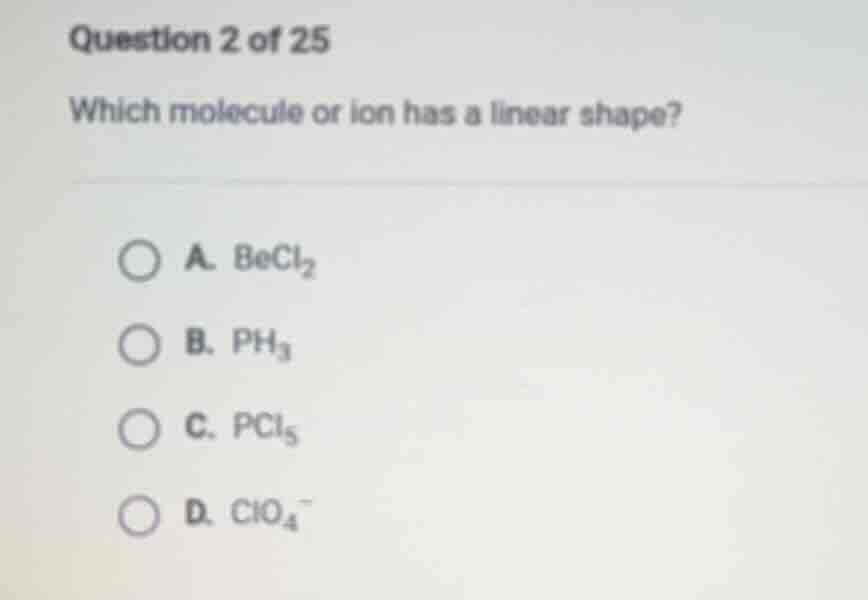
question 2 of 25
which molecule or ion has a linear shape?
a. becl₂
b. ph₃
c. pcl₅
d. clo₄⁻
To determine the molecular shape, we use the VSEPR theory. For \( \text{BeCl}_2 \): Be has 2 valence electrons, and each Cl contributes 1, so total valence electrons around Be is 2 (from bonding). The steric number (number of bonding pairs + lone pairs) for Be in \( \text{BeCl}_2 \) is 2 (no lone pairs on Be), so the shape is linear. For \( \text{PH}_3 \), P has a lone pair and 3 bonding pairs, giving a trigonal pyramidal shape. \( \text{PCl}_5 \) has a trigonal bipyramidal shape. \( \text{ClO}_4^- \) has a tetrahedral shape.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \( \text{BeCl}_2 \)