QUESTION IMAGE
Question
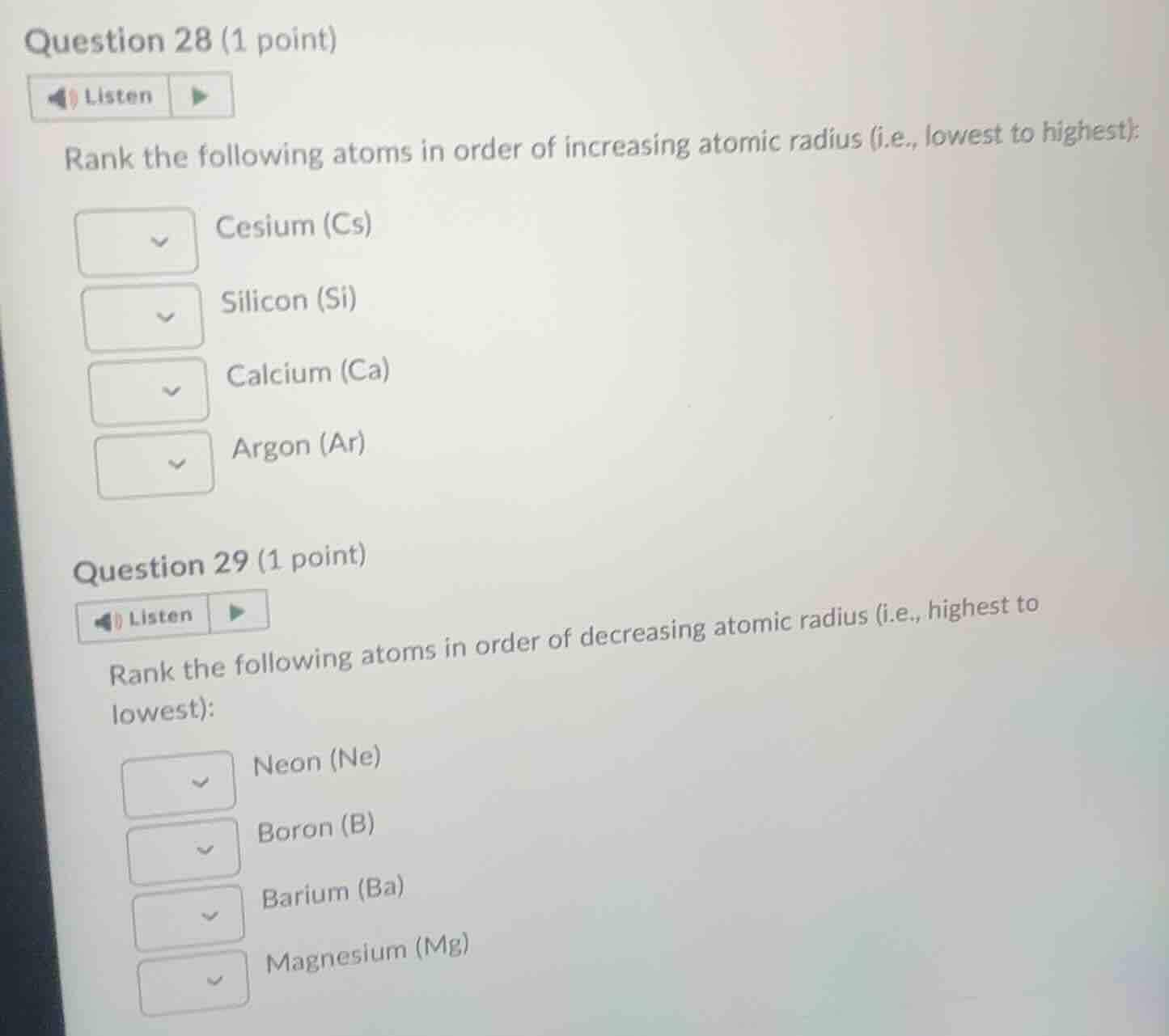
question 28 (1 point)listenrank the following atoms in order of increasing atomic radius (i.e., lowest to highest):cesium (cs)silicon (si)calcium (ca)argon (ar)question 29 (1 point)listenrank the following atoms in order of decreasing atomic radius (i.e., highest to lowest):neon (ne)boron (b)barium (ba)magnesium (mg)
For atomic radius trends: Across a period (left to right), atomic radius decreases as nuclear charge increases, pulling electrons closer. Down a group (top to bottom), atomic radius increases as new electron shells are added.
Question 28:
- Argon (Ar) is a noble gas in period 3, group 18 (smallest here).
- Silicon (Si) is in period 3, group 14 (larger than Ar, smaller than Ca).
- Calcium (Ca) is in period 4, group 2 (larger than Si, smaller than Cs).
- Cesium (Cs) is in period 6, group 1 (largest here).
Question 29:
- Barium (Ba) is in period 6, group 2 (largest here).
- Magnesium (Mg) is in period 3, group 2 (smaller than Ba, larger than B).
- Boron (B) is in period 2, group 13 (smaller than Mg, larger than Ne).
- Neon (Ne) is a noble gas in period 2, group 18 (smallest here).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 28 (increasing atomic radius, lowest to highest):
- Argon (Ar)
- Silicon (Si)
- Calcium (Ca)
- Cesium (Cs)
Question 29 (decreasing atomic radius, highest to lowest):
- Barium (Ba)
- Magnesium (Mg)
- Boron (B)
- Neon (Ne)