QUESTION IMAGE
Question
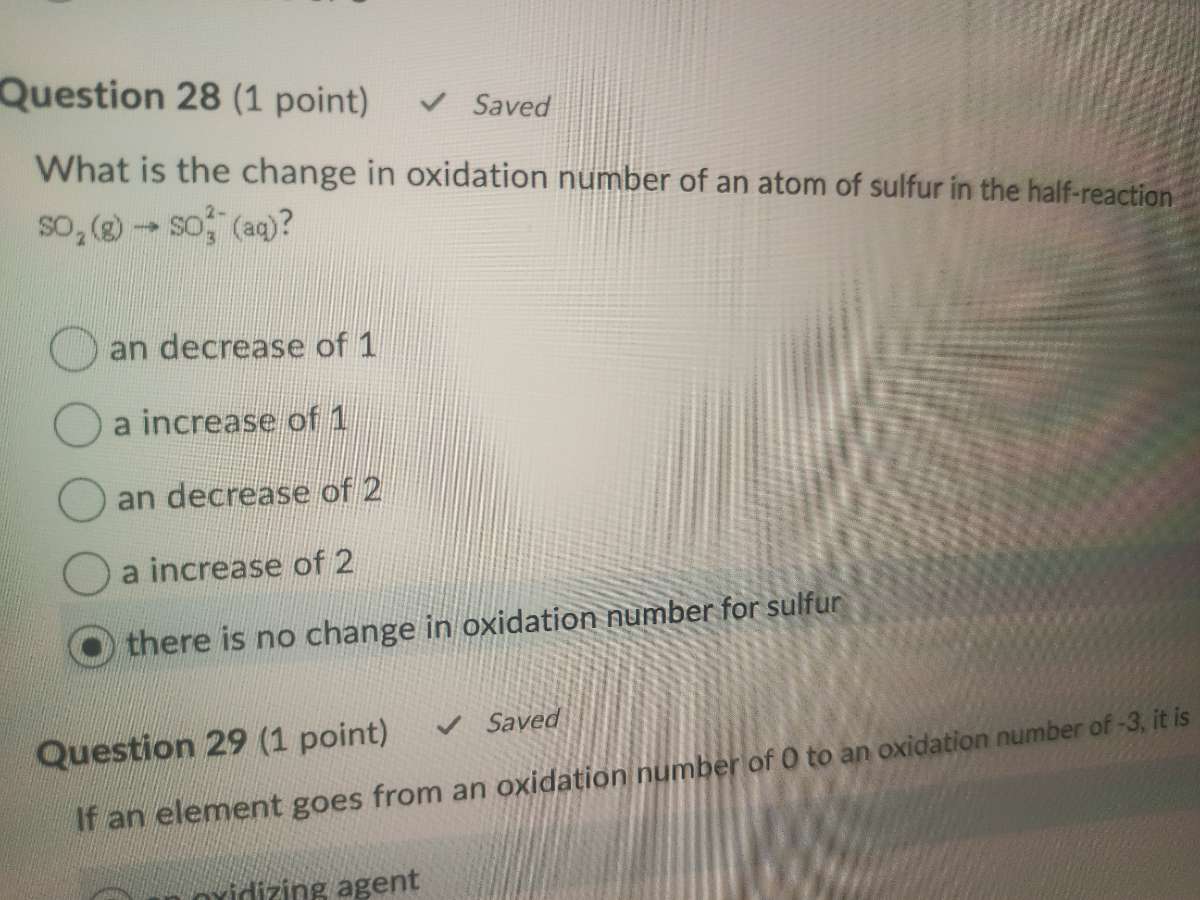
question 28 (1 point) saved
what is the change in oxidation number of an atom of sulfur in the half - reaction
$so_2(g)\
ightarrow so_3^{2 - }(aq)$?
\bigcirc an decrease of 1
\bigcirc a increase of 1
\bigcirc an decrease of 2
\bigcirc a increase of 2
\bigcirc there is no change in oxidation number for sulfur
question 29 (1 point) saved
if an element goes from an oxidation number of 0 to an oxidation number of - 3, it is
\bigcirc an oxidizing agent
Question 28
Step 1: Find oxidation number in \( \ce{SO2} \)
Oxygen has an oxidation number of -2. Let the oxidation number of S be \( x \). In \( \ce{SO2} \), we have \( x + 2(-2) = 0 \). Solving, \( x - 4 = 0 \) so \( x = +4 \).
Step 2: Find oxidation number in \( \ce{SO3^{2-}} \)
Oxygen is -2, charge is -2. Let S be \( y \). So \( y + 3(-2) = -2 \). Solving, \( y - 6 = -2 \) so \( y = +4 \).
Step 3: Compare oxidation numbers
Both are +4, so no change.
Oxidation number decreases (0 to -3, gain of electrons, reduction). A substance that is reduced is an oxidizing agent (it oxidizes other substances by accepting electrons).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
there is no change in oxidation number for sulfur
Question 29 (incomplete, but for the given part about oxidation number 0 to -3):
When an element's oxidation number decreases (from 0 to -3, it's a decrease), it gains electrons and is reduced. The substance that is reduced is the oxidizing agent. So if the question is about what it is (e.g., oxidizing agent), the answer would relate to that. But since the question is cut off, assuming the options include "an oxidizing agent" (or similar), the reasoning is: oxidation number decrease (reduction) → the element is reduced, so the substance is an oxidizing agent (as it causes oxidation of others by being reduced itself). If we assume the question is "If an element goes from an oxidation number of 0 to an oxidation number of -3, it is...", and options include "an oxidizing agent", then: