QUESTION IMAGE
Question
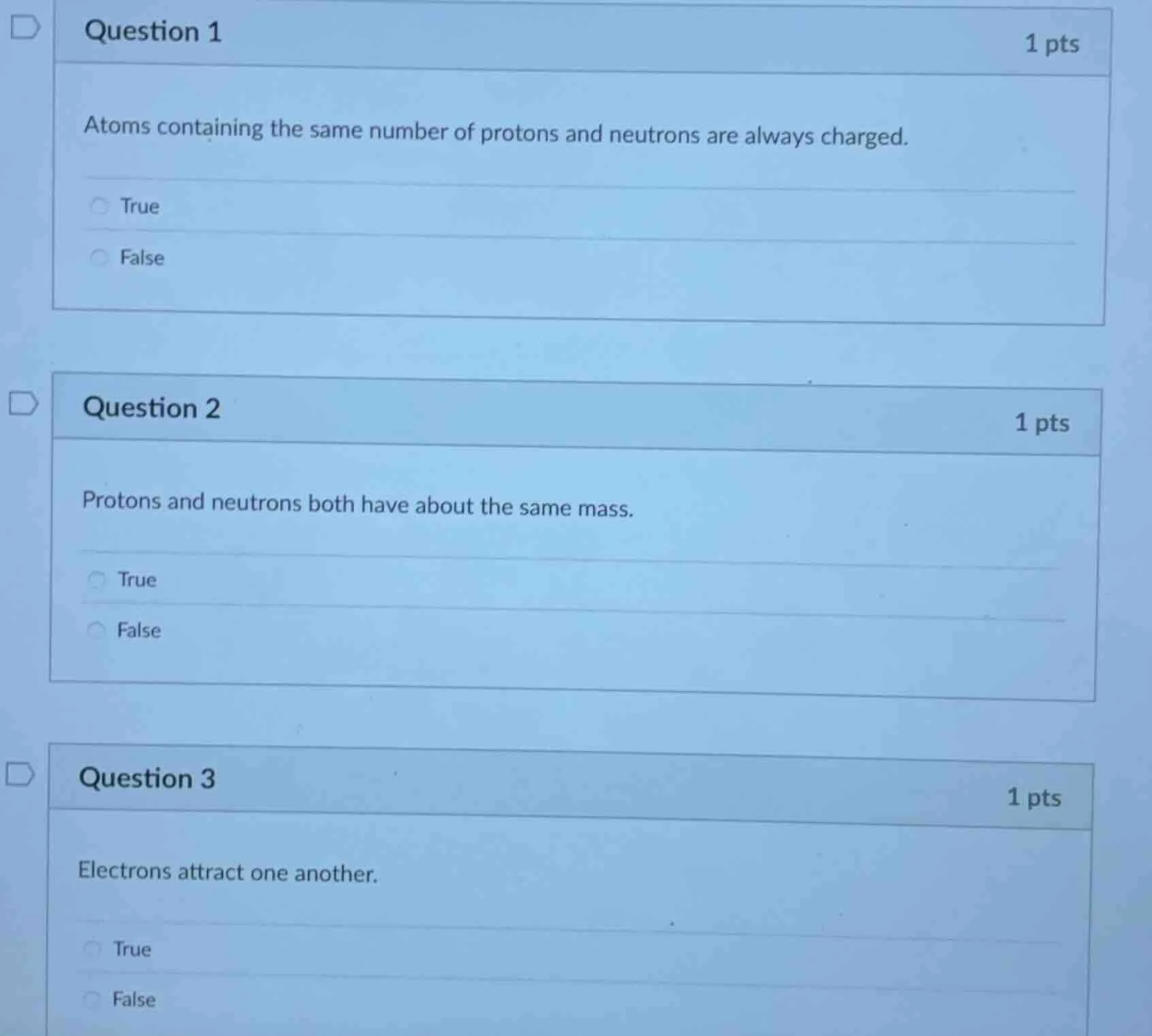
question 1
atoms containing the same number of protons and neutrons are always charged.
true
false
question 2
protons and neutrons both have about the same mass.
true
false
question 3
electrons attract one another.
true
false
Question 1
An atom's charge depends on the balance of protons (positive charge) and electrons (negative charge), not protons and neutrons. Atoms with equal protons and neutrons can be neutral (if electrons equal protons) or charged (ions), but "always charged" is false.
Protons and neutrons have masses of approximately \(1.67\times10^{-27}\) kg (proton) and \(1.675\times10^{-27}\) kg (neutron), which are about the same. So the statement is true.
Electrons have a negative charge. Like charges repel, so electrons repel each other, not attract. Thus the statement is false.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
False