QUESTION IMAGE
Question
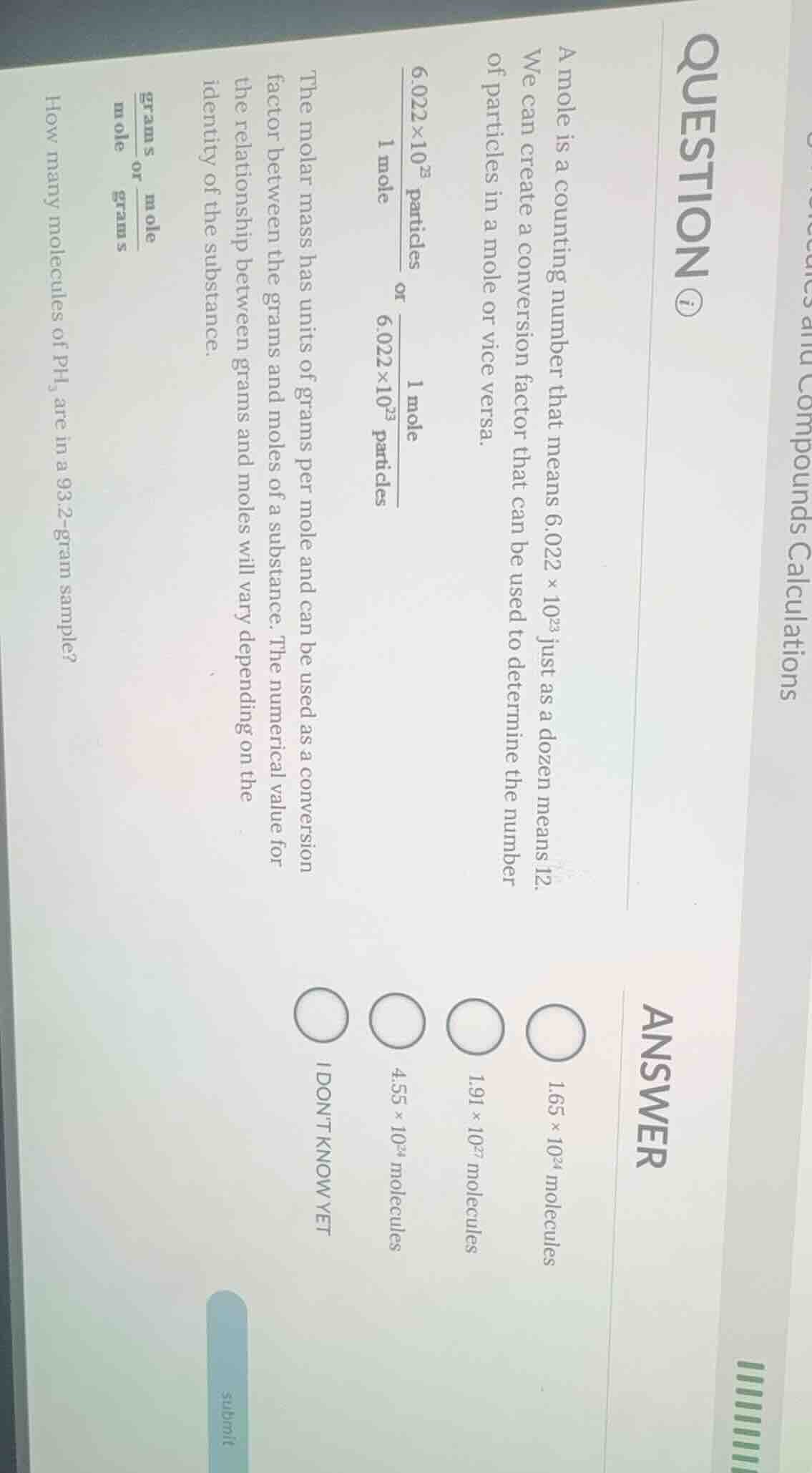
question ⓘcompounds calculationsa mole is a counting number that means $6.022 \times 10^{23}$ just as a dozen means 12.we can create a conversion factor that can be used to determine the number of particles in a mole or vice versa.$\frac{6.022 \times 10^{23}\text{ particles}}{1\text{ mole}}$ or $\frac{1\text{ mole}}{6.022 \times 10^{23}\text{ particles}}$the molar mass has units of grams per mole and can be used as a conversion factor between the grams and moles of a substance. the numerical value for the relationship between grams and moles will vary depending on the identity of the substance.$\frac{\text{grams}}{\text{mole}}$ or $\frac{\text{mole}}{\text{grams}}$how many molecules of $ph_3$ are in a 93.2-gram sample?answer$1.65 \times 10^{24}$ molecules$1.91 \times 10^{27}$ molecules$4.55 \times 10^{24}$ moleculesi dont know yet
Step1: Calculate molar mass of $\text{PH}_3$
Molar mass of $\text{P} = 30.97\ \text{g/mol}$, molar mass of $\text{H} = 1.008\ \text{g/mol}$
$$\text{Molar mass of }\text{PH}_3 = 30.97 + 3\times1.008 = 34.00\ \text{g/mol}$$
Step2: Convert grams to moles
$$\text{Moles of }\text{PH}_3 = \frac{93.2\ \text{g}}{34.00\ \text{g/mol}} = 2.741\ \text{mol}$$
Step3: Convert moles to molecules
$$\text{Number of molecules} = 2.741\ \text{mol} \times 6.022\times10^{23}\ \frac{\text{molecules}}{\text{mol}}$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1.65 × 10²⁴ molecules