QUESTION IMAGE
Question
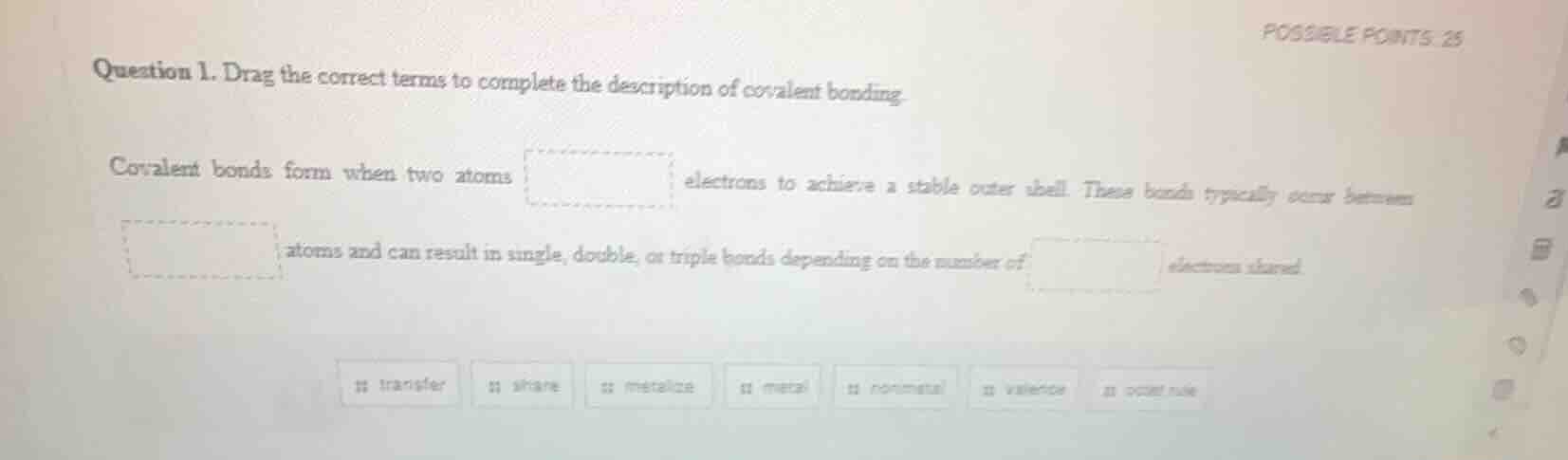
question 1. drag the correct terms to complete the description of covalent bonding.
covalent bonds form when two atoms
electrons to achieve a stable outer shell. these bonds typically occur between
atoms and can result in single, double, or triple bonds depending on the number of
electrons shared.
transfer share metallic metal nonmetal valence positive
Brief Explanations
To solve this, we recall the properties of covalent bonding:
- Covalent bonds involve atoms sharing electrons (ionic bonds transfer electrons).
- Covalent bonds typically occur between nonmetal atoms (metals form metallic bonds or ionic bonds with nonmetals).
- The number of bonds (single, double, triple) depends on the number of valence electrons shared (valence electrons are the outer - shell electrons involved in bonding).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First blank: share
Second blank: nonmetal
Third blank: valence