QUESTION IMAGE
Question
question 7
mark as: none
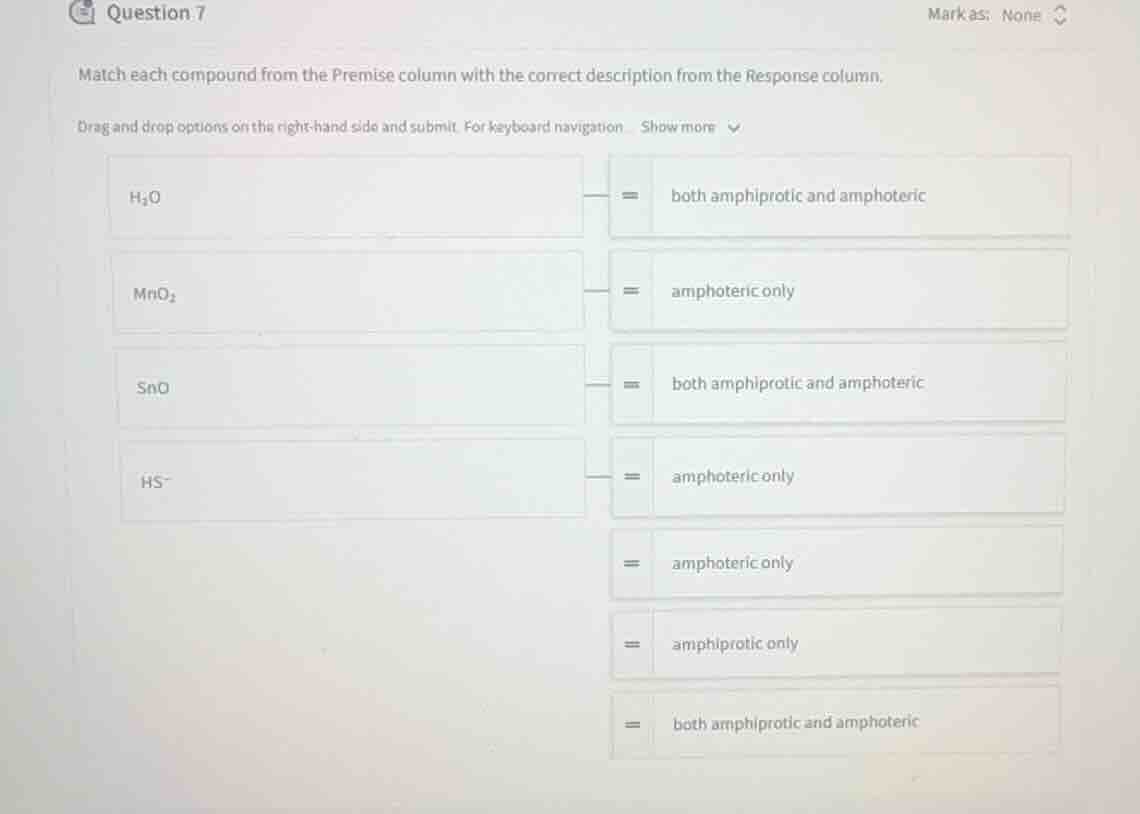
match each compound from the premise column with the correct description from the response column.
drag and drop options on the right-hand side and submit. for keyboard navigation show more
$h_2o$
both amphiprotic and amphoteric
$mno_2$
amphoteric only
$sno$
both amphiprotic and amphoteric
$hs^-$
amphoteric only
amphoteric only
amphiprotic only
both amphiprotic and amphoteric
- H₂O: Can donate a proton (acts as Brønsted acid: $\text{H}_2\text{O}
ightarrow \text{H}^+ + \text{OH}^-$) and accept a proton (acts as Brønsted base: $\text{H}_2\text{O} + \text{H}^+
ightarrow \text{H}_3\text{O}^+$), so it is amphiprotic. It also reacts with both strong acids and strong bases (e.g., with $\text{HCl}$ to form $\text{H}_3\text{O}^+$, with $\text{Na}$ to form $\text{NaOH}$), so it is amphoteric.
- MnO₂: Is a metal oxide that reacts with both strong acids (e.g., $\text{MnO}_2 + 4\text{HCl}
ightarrow \text{MnCl}_2 + \text{Cl}_2 + 2\text{H}_2\text{O}$) and strong bases (in fused conditions, $\text{MnO}_2 + 2\text{NaOH}
ightarrow \text{Na}_2\text{MnO}_3 + \text{H}_2\text{O}$), but it cannot donate or accept protons, so it is amphoteric only.
- SnO: Is a metal oxide that reacts with strong acids ($\text{SnO} + 2\text{HCl}
ightarrow \text{SnCl}_2 + \text{H}_2\text{O}$) and strong bases ($\text{SnO} + 2\text{NaOH}
ightarrow \text{Na}_2\text{SnO}_2 + \text{H}_2\text{O}$), but it has no protons to donate, so it is amphoteric only.
- HS⁻: Can donate a proton ($\text{HS}^-
ightarrow \text{H}^+ + \text{S}^{2-}$) and accept a proton ($\text{HS}^- + \text{H}^+
ightarrow \text{H}_2\text{S}$), so it is amphiprotic. It also acts as an amphoteric species by reacting with both strong acids and bases, so it is both amphiprotic and amphoteric.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{H}_2\text{O}$ = both amphiprotic and amphoteric
- $\text{MnO}_2$ = amphoteric only
- $\text{SnO}$ = amphoteric only
- $\text{HS}^-$ = both amphiprotic and amphoteric