QUESTION IMAGE
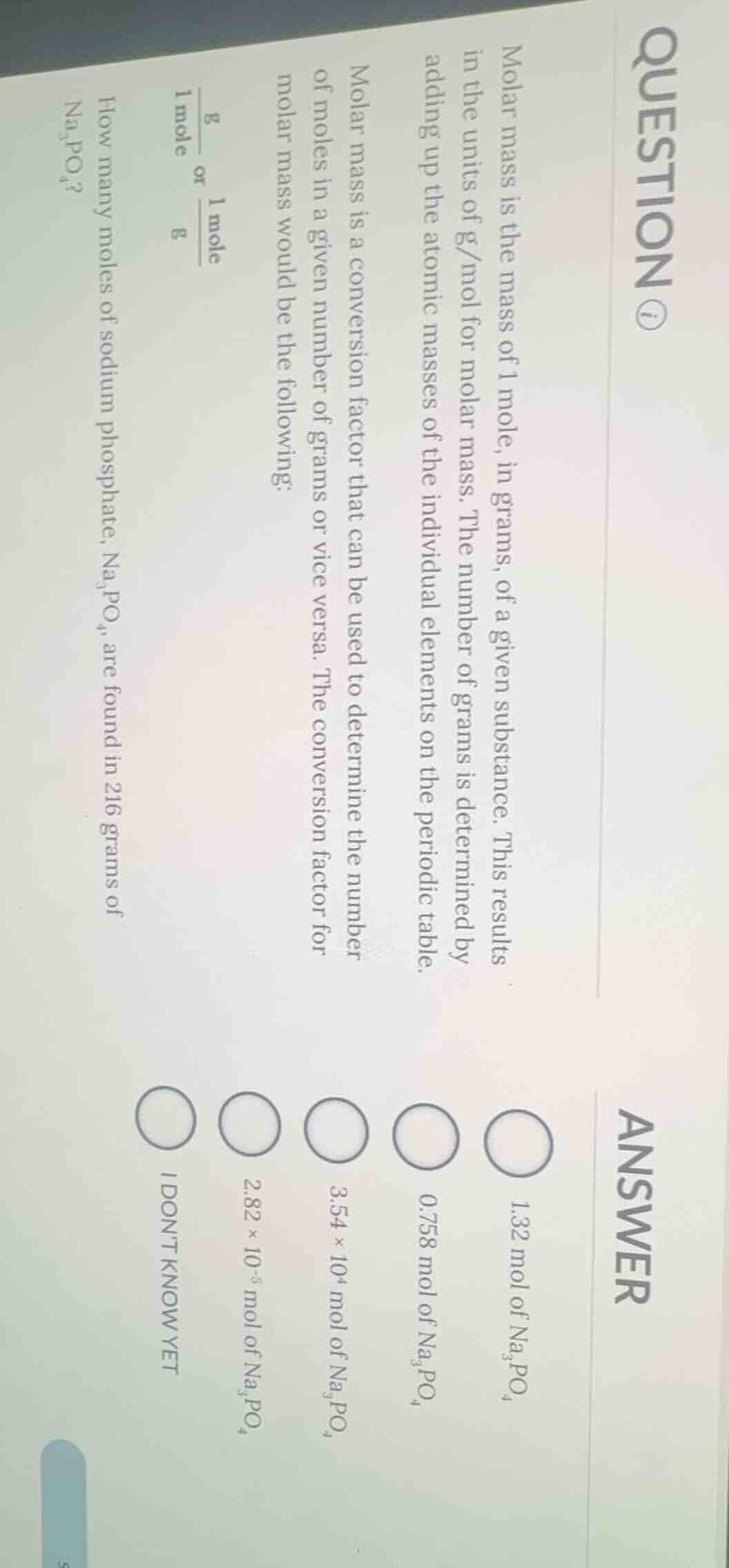
Question
question ⓘ
molar mass is the mass of 1 mole, in grams, of a given substance. this results in the units of g/mol for molar mass. the number of grams is determined by adding up the atomic masses of the individual elements on the periodic table.
molar mass is a conversion factor that can be used to determine the number of moles in a given number of grams or vice versa. the conversion factor for molar mass would be the following:
$\frac{\text{g}}{1\text{ mole}}$ or $\frac{1\text{ mole}}{\text{g}}$
how many moles of sodium phosphate, $\text{na}_3\text{po}_4$, are found in 216 grams of $\text{na}_3\text{po}_4$?
answer
$\bigcirc$ 1.32 mol of $\text{na}_3\text{po}_4$
$\bigcirc$ 0.758 mol of $\text{na}_3\text{po}_4$
$\bigcirc$ $3.54 \times 10^4$ mol of $\text{na}_3\text{po}_4$
$\bigcirc$ $2.82 \times 10^{-5}$ mol of $\text{na}_3\text{po}_4$
$\bigcirc$ i dont know yet
Step1: Calculate molar mass of $\text{Na}_3\text{PO}_4$
Find atomic masses: $\text{Na}=22.99\ \text{g/mol}$, $\text{P}=30.97\ \text{g/mol}$, $\text{O}=16.00\ \text{g/mol}$
$$\text{Molar mass} = (3 \times 22.99) + 30.97 + (4 \times 16.00) = 68.97 + 30.97 + 64.00 = 163.94\ \text{g/mol}$$
Step2: Convert grams to moles
Use formula: $\text{Moles} = \frac{\text{Mass}}{\text{Molar Mass}}$
$$\text{Moles of }\text{Na}_3\text{PO}_4 = \frac{216\ \text{g}}{163.94\ \text{g/mol}} \approx 1.32\ \text{mol}$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1.32 mol of $\text{Na}_3\text{PO}_4$