QUESTION IMAGE
Question
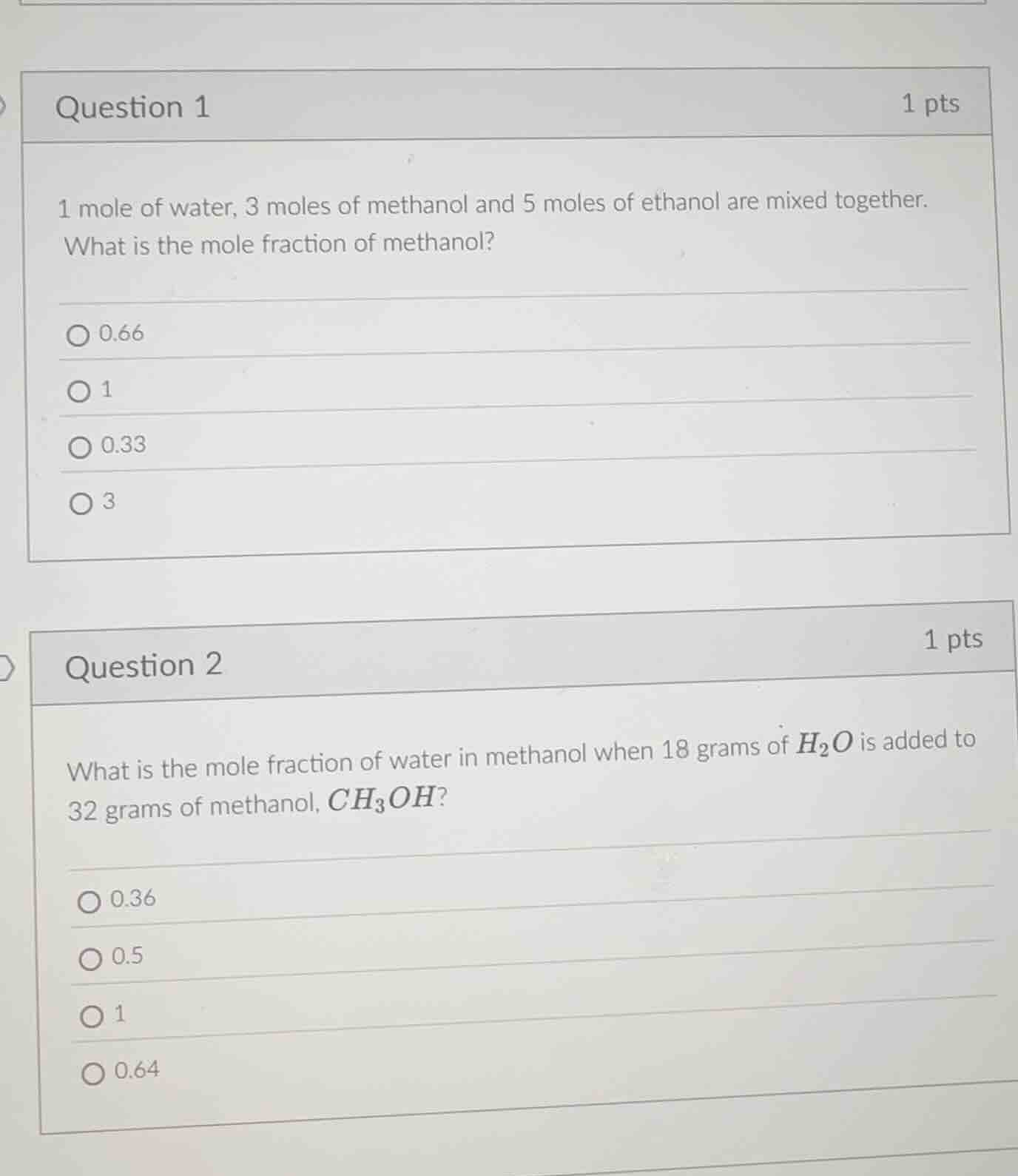
question 1
1 mole of water, 3 moles of methanol and 5 moles of ethanol are mixed together. what is the mole fraction of methanol?
0.66
1
0.33
3
question 2
what is the mole fraction of water in methanol when 18 grams of h₂o is added to 32 grams of methanol, ch₃oh?
0.36
0.5
1
0.64
Question 1
Step1: Calculate total moles
Total moles = moles of water + moles of methanol + moles of ethanol = \(1 + 3 + 5 = 9\) moles.
Step2: Calculate mole fraction of methanol
Mole fraction of methanol (\(X_{methanol}\)) = \(\frac{\text{moles of methanol}}{\text{total moles}}\) = \(\frac{3}{9}=\frac{1}{3}\approx0.33\).
Step1: Calculate moles of \(H_2O\)
Molar mass of \(H_2O\) is \(18\space g/mol\). Moles of \(H_2O\) = \(\frac{\text{mass}}{\text{molar mass}}=\frac{18\space g}{18\space g/mol}=1\space mol\).
Step2: Calculate moles of \(CH_3OH\)
Molar mass of \(CH_3OH\) is \(32\space g/mol\) (C:12, H:4×1, O:16; \(12 + 4+16 = 32\)). Moles of \(CH_3OH\) = \(\frac{32\space g}{32\space g/mol}=1\space mol\).
Step3: Calculate total moles
Total moles = moles of \(H_2O\) + moles of \(CH_3OH\) = \(1 + 1 = 2\space mol\).
Step4: Calculate mole fraction of \(H_2O\)
Mole fraction of \(H_2O\) (\(X_{H_2O}\)) = \(\frac{\text{moles of }H_2O}{\text{total moles}}=\frac{1}{2}=0.5\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0.33