QUESTION IMAGE
Question
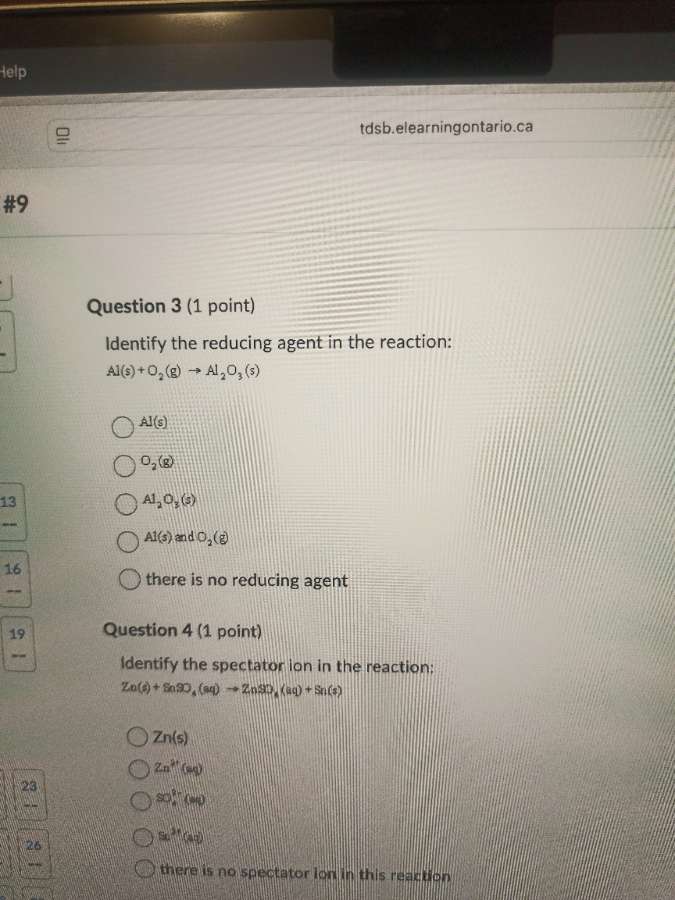
question 3 (1 point)
identify the reducing agent in the reaction:
al(s) + o₂(g) → al₂o₃(s)
al(s)
o₂(g)
al₂o₃(s)
al(s) and o₂(g)
there is no reducing agent
question 4 (1 point)
identify the spectator ion in the reaction:
zn(s) + snso₄(aq) → znso₄(aq) + sn(s)
zn(s)
zn²⁺(aq)
so₄²⁻(aq)
sn²⁺(aq)
there is no spectator ion in this reaction
Question 3
To identify the reducing agent, we look at oxidation states. In \( \text{Al}(s) + \text{O}_2(g)
ightarrow \text{Al}_2\text{O}_3(s) \), Al goes from 0 (in Al(s)) to +3 (in \( \text{Al}_2\text{O}_3 \)), so it is oxidized. The substance that is oxidized is the reducing agent. So Al(s) is the reducing agent.
For the reaction \( \text{Zn}(s) + \text{SnSO}_4(aq)
ightarrow \text{ZnSO}_4(aq) + \text{Sn}(s) \), we write the ionic equation. \( \text{SnSO}_4 \) dissociates into \( \text{Sn}^{2+}(aq) \) and \( \text{SO}_4^{2-}(aq) \), \( \text{ZnSO}_4 \) dissociates into \( \text{Zn}^{2+}(aq) \) and \( \text{SO}_4^{2-}(aq) \). The ionic reaction is \( \text{Zn}(s) + \text{Sn}^{2+}(aq)
ightarrow \text{Zn}^{2+}(aq) + \text{Sn}(s) \). The \( \text{SO}_4^{2-} \) ion does not participate in the reaction (remains the same on both sides), so it is the spectator ion.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \( \text{Al}(s) \)