QUESTION IMAGE
Question
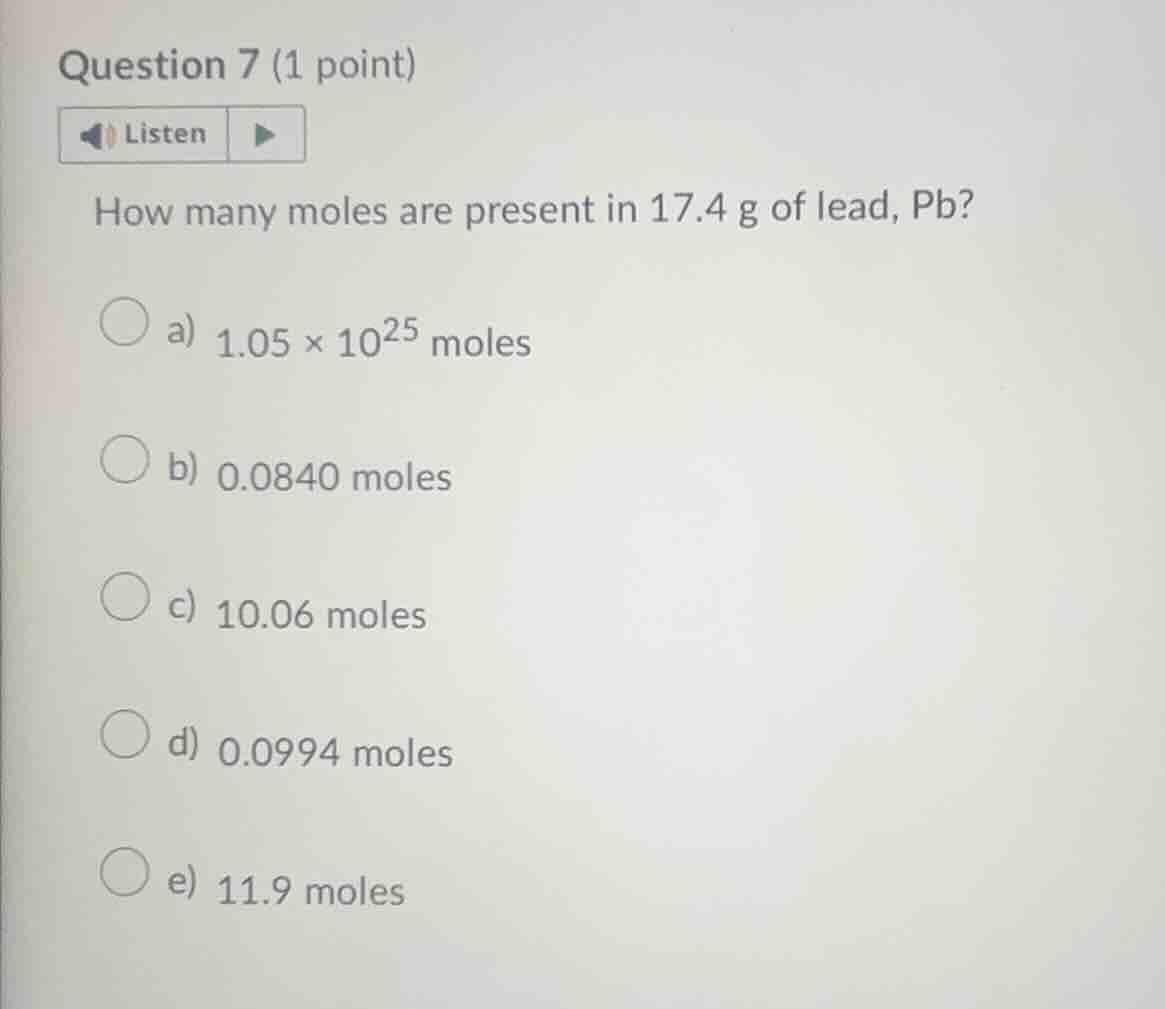
question 7 (1 point) listen how many moles are present in 17.4 g of lead, pb? a) $1.05 \times 10^{25}$ moles b) 0.0840 moles c) 10.06 moles d) 0.0994 moles e) 11.9 moles
Step1: Find molar mass of Pb
Molar mass of Pb = $207.2\ \text{g/mol}$
Step2: Calculate moles via mass/molar mass
$\text{Moles of Pb} = \frac{17.4\ \text{g}}{207.2\ \text{g/mol}}$
Step3: Compute final value
$\text{Moles of Pb} \approx 0.0840\ \text{moles}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
b) 0.0840 moles