QUESTION IMAGE
Question
question 5 (0.5 points)
listen
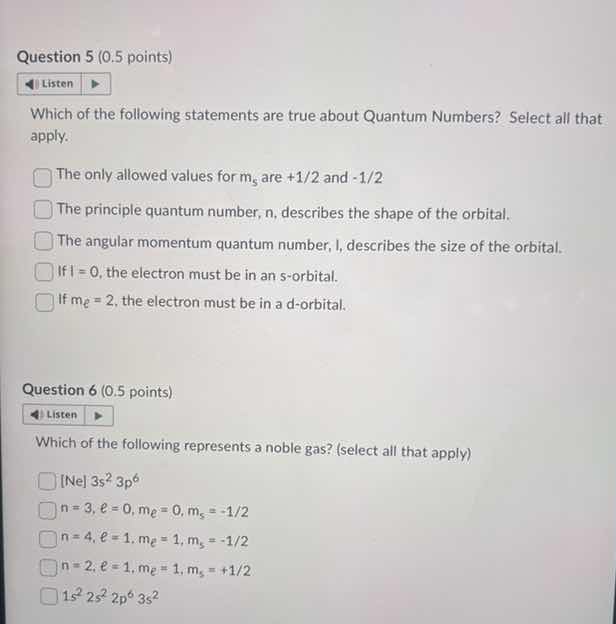
which of the following statements are true about quantum numbers? select all that apply.
the only allowed values for $m_s$ are +1/2 and -1/2
the principle quantum number, n, describes the shape of the orbital.
the angular momentum quantum number, l, describes the size of the orbital.
if l = 0, the electron must be in an s - orbital.
if $m_l$ = 2, the electron must be in a d - orbital.
question 6 (0.5 points)
listen
which of the following represents a noble gas? (select all that apply)
ne $3s^2 3p^6$
$n = 3, \ell = 0, m_\ell = 0, m_s = -1/2$
$n = 4, \ell = 1, m_\ell = 1, m_s = -1/2$
$n = 2, \ell = 1, m_\ell = 1, m_s = +1/2$
$1s^2 2s^2 2p^6 3s^2$
Question 5
- $m_s$ (spin quantum number) only has two allowed values: $+1/2$ and $-1/2$.
- The principal quantum number $n$ describes orbital size, not shape.
- The angular momentum quantum number $l$ describes orbital shape, not size.
- When $l=0$, the orbital type is always an s-orbital.
- For $m_l=2$, the minimum $l$ value is 2 (since $m_l$ ranges from $-l$ to $+l$), which corresponds to d-orbitals.
A noble gas has a full valence electron shell (ns²np⁶ for main group noble gases, or a filled core for heavier ones).
- $[Ne] 3s^2 3p^6$ is the electron configuration of argon, a noble gas.
- $n=3, l=0, m_l=0, m_s=-1/2$ describes a single electron in a 3s orbital, not a full noble gas configuration.
- $n=4, l=1, m_l=1, m_s=-1/2$ describes a single electron in a 4p orbital, not a full noble gas configuration.
- $n=2, l=1, m_l=1, m_s=+1/2$ describes a single electron in a 2p orbital, not a full noble gas configuration.
- $1s^2 2s^2 2p^6 3s^2$ has an incomplete 3p subshell, so it is magnesium, not a noble gas.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- The only allowed values for $m_s$ are +1/2 and -1/2
- If $l = 0$, the electron must be in an s-orbital.
- If $m_l = 2$, the electron must be in a d-orbital.
---