QUESTION IMAGE
Question
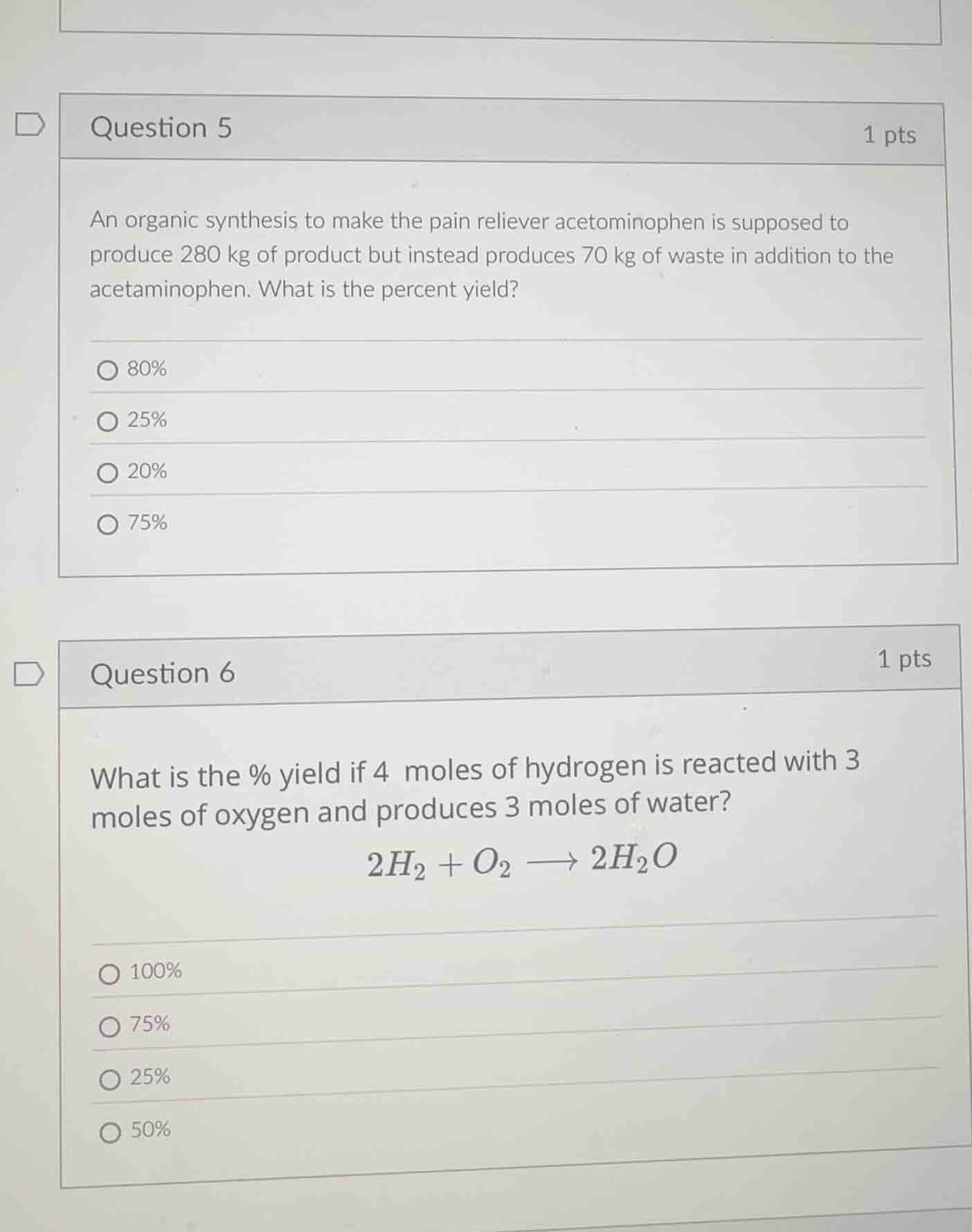
question 5
1 pts
an organic synthesis to make the pain reliever acetominophen is supposed to produce 280 kg of product but instead produces 70 kg of waste in addition to the acetaminophen. what is the percent yield?
80%
25%
20%
75%
question 6
1 pts
what is the % yield if 4 moles of hydrogen is reacted with 3 moles of oxygen and produces 3 moles of water?
2h₂ + o₂ → 2h₂o
100%
75%
25%
50%
Question 5
Step1: Find actual product mass
The total mass of product (acetaminophen) and waste is the theoretical mass? Wait, no. Wait, the theoretical yield is 280 kg, but instead, we have 70 kg waste plus acetaminophen. Wait, no—wait, the theoretical yield is the expected product mass. Wait, maybe the total mass of reactants would be product + waste? Wait, no, percent yield is (actual yield / theoretical yield) 100. Wait, maybe the actual product mass is theoretical - waste? Wait, no, the problem says "supposed to produce 280 kg of product but instead produces 70 kg of waste in addition to the acetaminophen". So total mass from reaction is product (acetaminophen) + waste = 280 kg? Wait, no—wait, maybe the theoretical yield is 280 kg (product), but the actual product is 280 - 70 = 210 kg? Wait, no, that doesn't make sense. Wait, no—wait, percent yield is (actual yield / theoretical yield) 100. Let's re-read: "An organic synthesis to make the pain reliever acetominophen is supposed to produce 280 kg of product but instead produces 70 kg of waste in addition to the acetaminophen." So "supposed to produce 280 kg of product" (theoretical yield is 280 kg product). But instead, they have 70 kg waste + acetaminophen. Wait, maybe the total mass of product + waste is equal to the theoretical product? No, that's not right. Wait, maybe the theoretical yield is 280 kg (product), and the actual product is 280 - 70 = 210 kg? Wait, no, that would be if waste is subtracted from theoretical. Wait, no, percent yield is (actual / theoretical) 100. Let's think again. Wait, maybe the "supposed to produce 280 kg" is the theoretical yield (product), but the actual product is 280 - 70 = 210? No, that can't be. Wait, no—wait, maybe the total mass of product and waste is 280 kg? So product (actual) + waste (70 kg) = 280 kg. So actual product = 280 - 70 = 210 kg. Then percent yield is (210 / 280) 100 = 75%? Wait, no, 210/280 is 0.75, which is 75%. Wait, but let's check the options. 75% is an option. Wait, let's do the calculation:
Theoretical yield (product) = 280 kg.
Actual product = total (theoretical?) - waste? Wait, maybe the problem is that the theoretical yield is 280 kg (product), but the actual product is 280 - 70 = 210 kg. Then percent yield = (210 / 280) * 100 = 75%. So 75%.
Step2: Calculate percent yield
Percent yield = (actual yield / theoretical yield) * 100.
Actual yield (product) = 280 kg - 70 kg = 210 kg? Wait, no—wait, maybe the "supposed to produce 280 kg" is the total (product + waste), but that doesn't make sense. Wait, no, the problem says "supposed to produce 280 kg of product"—so theoretical yield is 280 kg (product). But instead, they have 70 kg waste + product. So product (actual) + 70 kg waste = 280 kg? So product (actual) = 280 - 70 = 210 kg. Then percent yield = (210 / 280) * 100 = 75%. Yes, that works. So 75%.
Step1: Determine limiting reactant
The reaction is \( 2H_2 + O_2
ightarrow 2H_2O \).
Moles of \( H_2 = 4 \), moles of \( O_2 = 3 \).
From the reaction, 2 moles \( H_2 \) react with 1 mole \( O_2 \) to produce 2 moles \( H_2O \).
For \( H_2 \): 4 moles \( H_2 \) would react with \( \frac{4}{2} = 2 \) moles \( O_2 \). We have 3 moles \( O_2 \), so \( H_2 \) is not limiting.
For \( O_2 \): 3 moles \( O_2 \) would react with \( 2 \times 3 = 6 \) moles \( H_2 \). But we have only 4 moles \( H_2 \), so \( H_2 \) is limiting (since we have less \( H_2 \) than needed for 3 moles \( O_2 \)).
Step2: Calculate theoretical yield of \( H_2O \)
From the reaction, 2 moles \( H_2 \) produce 2 moles \( H_2O \) (1:1 ratio for \( H_2 \) and \( H_2O \)).
Moles of \( H_2 = 4 \), so theoretical moles of \( H_2O = 4 \) (since 2 \( H_2 \) → 2 \( H_2O \), so 4 \( H_2 \) → 4 \( H_2O \)).
Step3: Calculate percent yield
Actual yield of \( H_2O = 3 \) moles.
Percent yield = \( \frac{\text{actual yield}}{\text{theoretical yield}} \times 100 = \frac{3}{4} \times 100 = 75\% \)? Wait, no—wait, wait, step 2: wait, 2 moles \( H_2 \) produce 2 moles \( H_2O \), so 4 moles \( H_2 \) would produce 4 moles \( H_2O \) (theoretical). Actual is 3 moles. So percent yield = (3/4)*100 = 75%? Wait, but let's check again.
Wait, reaction: 2 \( H_2 \) + 1 \( O_2 \) → 2 \( H_2O \).
Moles of \( H_2 = 4 \), moles of \( O_2 = 3 \).
Moles of \( H_2 \) needed for 3 moles \( O_2 \): 2*3 = 6. But we have 4, so \( H_2 \) is limiting. So moles of \( H_2O \) from \( H_2 \): 4 moles \( H_2 \) → 4 moles \( H_2O \) (since 2 \( H_2 \) → 2 \( H_2O \), so 1:1).
Actual moles \( H_2O = 3 \).
Percent yield = (3 / 4) * 100 = 75%.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
75% (Option D: 75%)