QUESTION IMAGE
Question
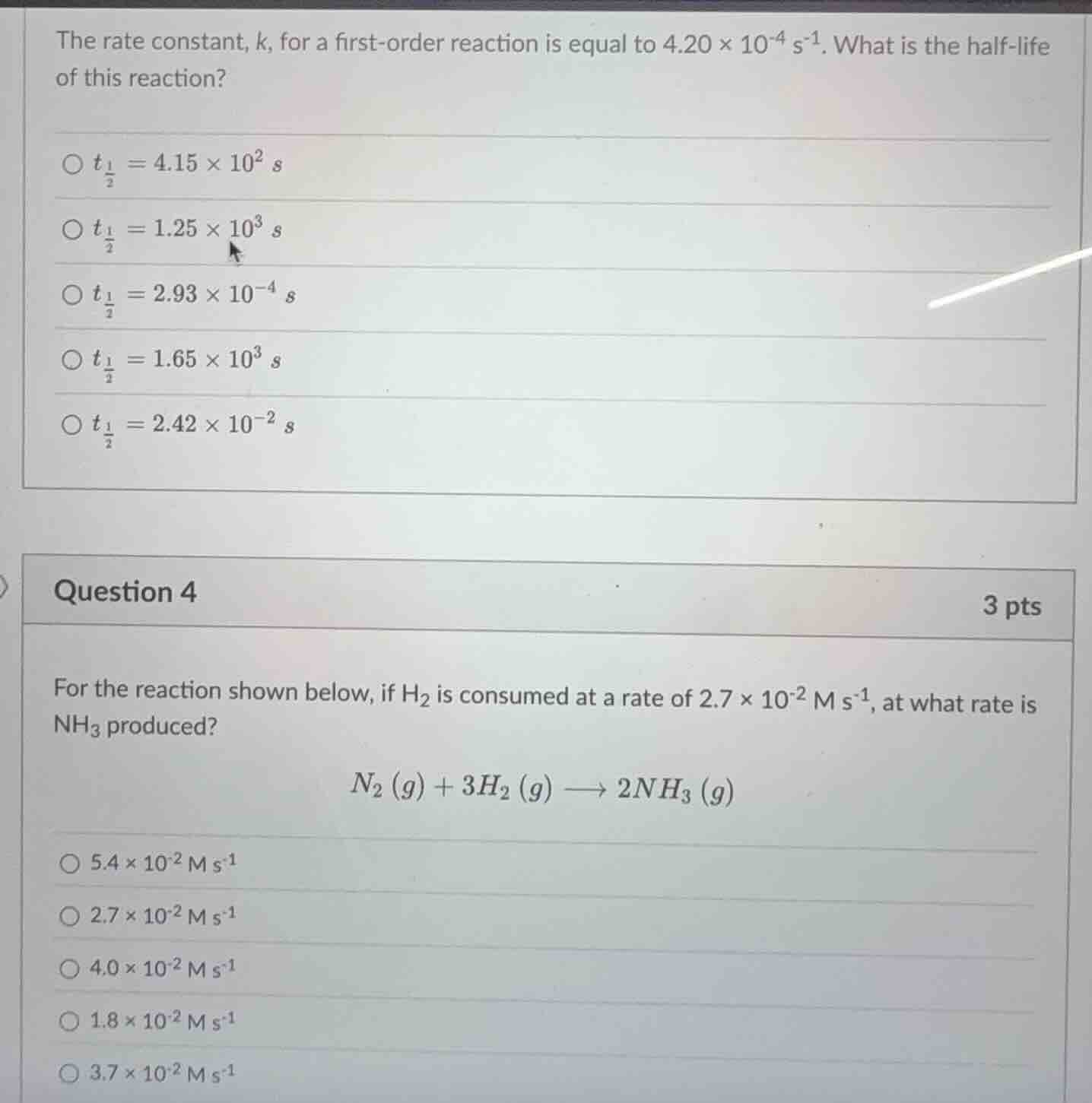
the rate constant, k, for a first-order reaction is equal to 4.20 × 10⁻⁴ s⁻¹. what is the half-life of this reaction?
○ $t_{\frac{1}{2}} = 4.15 × 10² s$
○ $t_{\frac{1}{2}} = 1.25 × 10³ s$
○ $t_{\frac{1}{2}} = 2.93 × 10⁻⁴ s$
○ $t_{\frac{1}{2}} = 1.65 × 10³ s$
○ $t_{\frac{1}{2}} = 2.42 × 10⁻² s$
question 4
3 pts
for the reaction shown below, if h₂ is consumed at a rate of 2.7 × 10⁻² m s⁻¹, at what rate is nh₃ produced?
$n₂ (g) + 3h₂ (g) \longrightarrow 2nh₃ (g)$
○ 5.4 × 10⁻² m s⁻¹
○ 2.7 × 10⁻² m s⁻¹
○ 4.0 × 10⁻² m s⁻¹
○ 1.8 × 10⁻² m s⁻¹
○ 3.7 × 10⁻² m s⁻¹
First Question (Half - life of first - order reaction)
Step1: Recall the formula for half - life of first - order reaction
The formula for the half - life (\(t_{\frac{1}{2}}\)) of a first - order reaction is \(t_{\frac{1}{2}}=\frac{\ln2}{k}\), where \(k\) is the rate constant and \(\ln2\approx0.693\).
Step2: Substitute the given value of \(k\)
We are given \(k = 4.20\times10^{-4}\ s^{-1}\). Substitute \(k\) into the formula:
\(t_{\frac{1}{2}}=\frac{0.693}{4.20\times 10^{-4}\ s^{-1}}\)
First, calculate \(\frac{0.693}{4.20\times10^{-4}}\):
\(0.693\div(4.20\times10^{-4})=\frac{0.693}{4.20}\times10^{4}\)
\(\frac{0.693}{4.20}\approx0.165\)
\(0.165\times10^{4}=1.65\times 10^{3}\ s\)
Step1: Recall the reaction rate relationship from stoichiometry
For the reaction \(N_{2}(g)+3H_{2}(g)
ightarrow2NH_{3}(g)\), the rate of consumption of \(H_{2}\) and the rate of production of \(NH_{3}\) are related by the stoichiometric coefficients. The rate relationship is \(\frac{\text{Rate of consumption of }H_{2}}{3}=\frac{\text{Rate of production of }NH_{3}}{2}\)
Step2: Solve for the rate of production of \(NH_{3}\)
Let \(r_{H_{2}}\) be the rate of consumption of \(H_{2}\) and \(r_{NH_{3}}\) be the rate of production of \(NH_{3}\). We know \(r_{H_{2}} = 2.7\times10^{-2}\ M\ s^{-1}\)
From \(\frac{r_{H_{2}}}{3}=\frac{r_{NH_{3}}}{2}\), we can re - arrange to get \(r_{NH_{3}}=\frac{2}{3}\times r_{H_{2}}\)
Substitute \(r_{H_{2}} = 2.7\times10^{-2}\ M\ s^{-1}\) into the formula:
\(r_{NH_{3}}=\frac{2}{3}\times(2.7\times 10^{-2}\ M\ s^{-1})\)
\(\frac{2}{3}\times2.7\times10^{-2}=1.8\times 10^{-2}\ M\ s^{-1}\)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(t_{\frac{1}{2}} = 1.65\times10^{3}\ s\) (the option: \(t_{\frac{1}{2}} = 1.65\times10^{3}\ s\))