QUESTION IMAGE
Question
(此处省略图像相关内容,仅保留选项相关文字:选项b、c、d,以及反应进程与势能的图像相关描述,如reactants、produ、reaction progress、potential energy等)
To determine the correct reaction energy diagram, we analyze the potential energy of reactants and products:
Key Concept:
- Exothermic reaction: Products have lower potential energy than reactants (energy is released).
- Endothermic reaction: Products have higher potential energy than reactants (energy is absorbed).
- All reactions have an activation energy (energy barrier) that reactants must overcome to form products.
Analyzing Each Option:
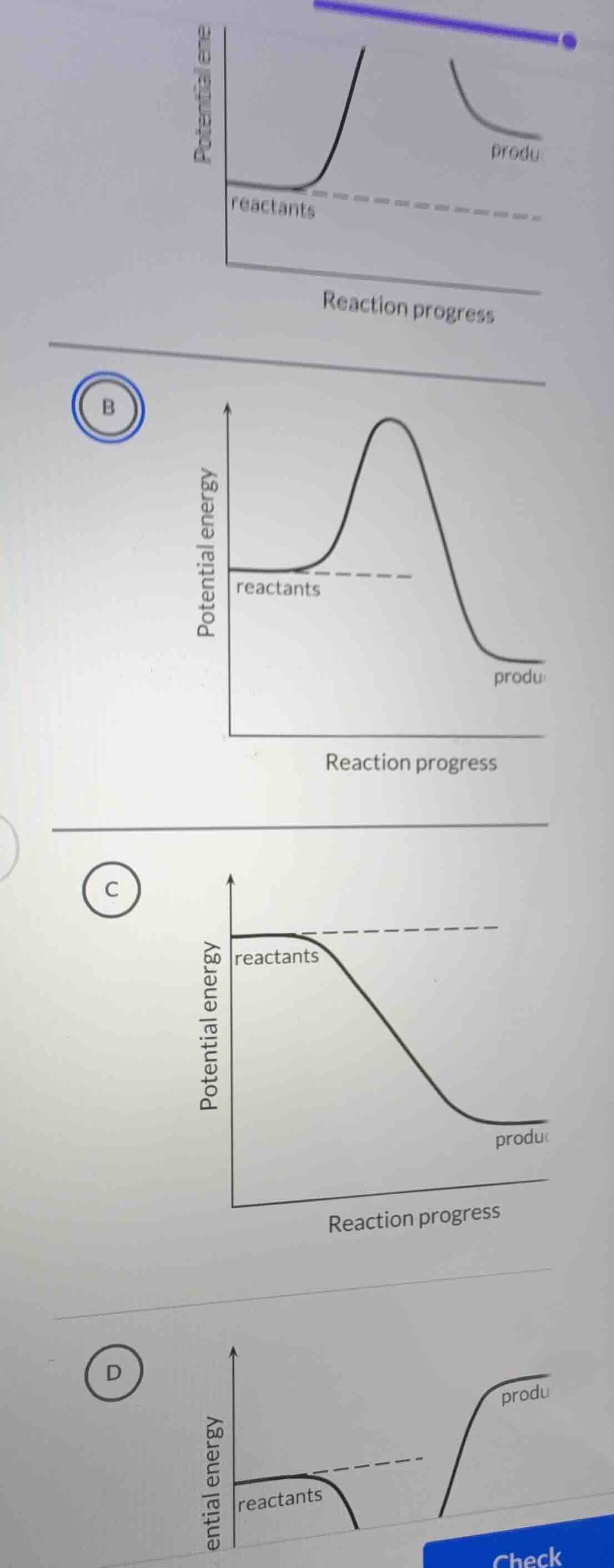
- Option A: Products have higher energy than reactants (endothermic), but the activation energy path is unclear (incomplete diagram).
- Option B: Reactants and products start at the same energy, but the product energy is lower (exothermic), and there is a clear activation energy barrier (peak) — consistent with a typical exothermic reaction profile.
- Option C: No activation energy barrier (incorrect, as all reactions have an activation energy).
- Option D: Products have higher energy than reactants (endothermic), but the diagram is inconsistent (e.g., reactants’ energy is lower, and the product path is unusual).
For a valid reaction energy diagram, there must be an activation energy barrier, and the energy change (reactants → products) must be consistent. Option B shows a clear activation energy peak and products with lower energy than reactants (exothermic), which is a standard reaction profile.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. (The diagram labeled B, with reactants at a certain energy, a peak (activation energy), and products at lower energy than reactants)