QUESTION IMAGE
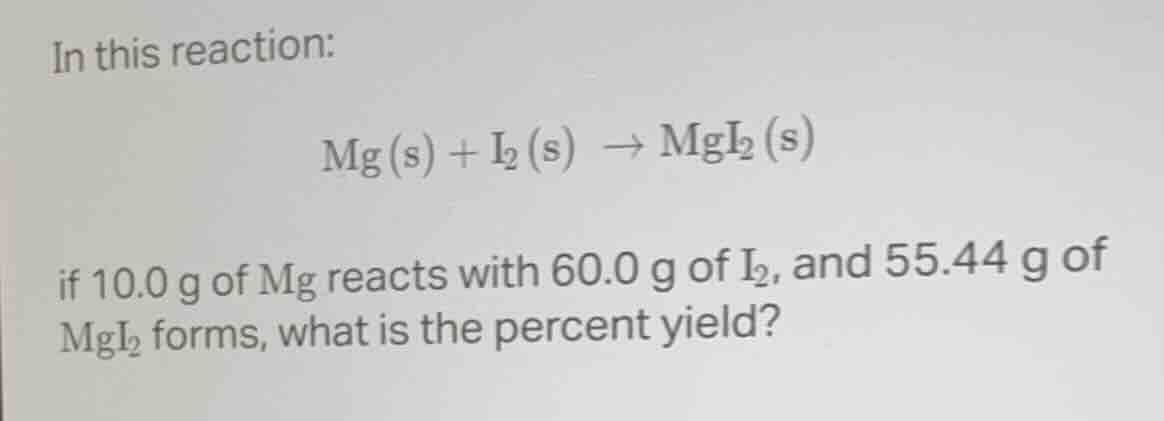
Question
in this reaction:
$ce{mg(s) + i_{2}(s) -> mgi_{2}(s)}$
if 10.0 g of mg reacts with 60.0 g of $ce{i_{2}}$, and 55.44 g of $ce{mgi_{2}}$ forms, what is the percent yield?
Step1: Calculate molar masses
Molar mass of $\text{Mg} = 24.305\ \text{g/mol}$
Molar mass of $\text{I}_2 = 2\times126.90 = 253.80\ \text{g/mol}$
Molar mass of $\text{MgI}_2 = 24.305 + 2\times126.90 = 278.105\ \text{g/mol}$
Step2: Find moles of reactants
Moles of $\text{Mg} = \frac{10.0\ \text{g}}{24.305\ \text{g/mol}} \approx 0.411\ \text{mol}$
Moles of $\text{I}_2 = \frac{60.0\ \text{g}}{253.80\ \text{g/mol}} \approx 0.236\ \text{mol}$
Step3: Identify limiting reactant
From reaction, 1 mol $\text{Mg}$ reacts with 1 mol $\text{I}_2$. $\text{I}_2$ has fewer moles, so it is limiting.
Step4: Calculate theoretical yield
1 mol $\text{I}_2$ produces 1 mol $\text{MgI}_2$, so moles of $\text{MgI}_2$ (theoretical) = 0.236 mol
Theoretical yield = $0.236\ \text{mol} \times 278.105\ \text{g/mol} \approx 65.6\ \text{g}$
Step5: Calculate percent yield
Percent yield = $\frac{\text{Actual Yield}}{\text{Theoretical Yield}} \times 100\%$
Percent yield = $\frac{55.44\ \text{g}}{65.6\ \text{g}} \times 100\%$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Approximately 84.5%