QUESTION IMAGE
Question
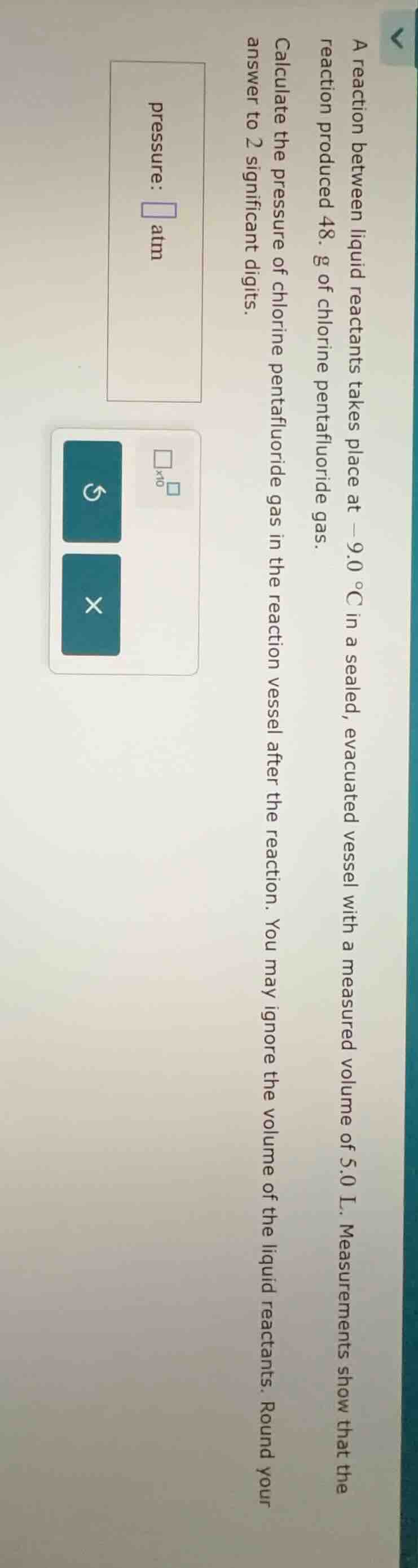
a reaction between liquid reactants takes place at -9.0 °c in a sealed, evacuated vessel with a measured volume of 5.0 l. measurements show that the reaction produced 48. g of chlorine pentafluoride gas. calculate the pressure of chlorine pentafluoride gas in the reaction vessel after the reaction. you may ignore the volume of the liquid reactants. round your answer to 2 significant digits. pressure: atm
Step1: Calculate moles of \( \text{ClF}_5 \)
Molar mass of \( \text{ClF}_5 \): \( \text{Cl} (35.45\,\text{g/mol}) + 5\times \text{F} (19.00\,\text{g/mol}) = 35.45 + 95.00 = 130.45\,\text{g/mol} \).
Moles (\( n \)): \( n = \frac{\text{mass}}{\text{molar mass}} = \frac{48.\,\text{g}}{130.45\,\text{g/mol}} \approx 0.368\,\text{mol} \).
Step2: Convert temperature to Kelvin
\( T = -9.0^\circ\text{C} + 273.15 = 264.15\,\text{K} \).
Step3: Apply Ideal Gas Law (\( PV = nRT \))
\( R = 0.0821\,\text{L·atm/(mol·K)} \), \( V = 5.0\,\text{L} \).
Solve for \( P \): \( P = \frac{nRT}{V} \).
Substitute values: \( P = \frac{0.368\,\text{mol} \times 0.0821\,\text{L·atm/(mol·K)} \times 264.15\,\text{K}}{5.0\,\text{L}} \).
Calculate numerator: \( 0.368 \times 0.0821 \times 264.15 \approx 0.368 \times 21.69 \approx 7.98 \).
Divide by \( V \): \( P \approx \frac{7.98}{5.0} \approx 1.6\,\text{atm} \) (2 significant digits).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 1.6 \)