QUESTION IMAGE
Question
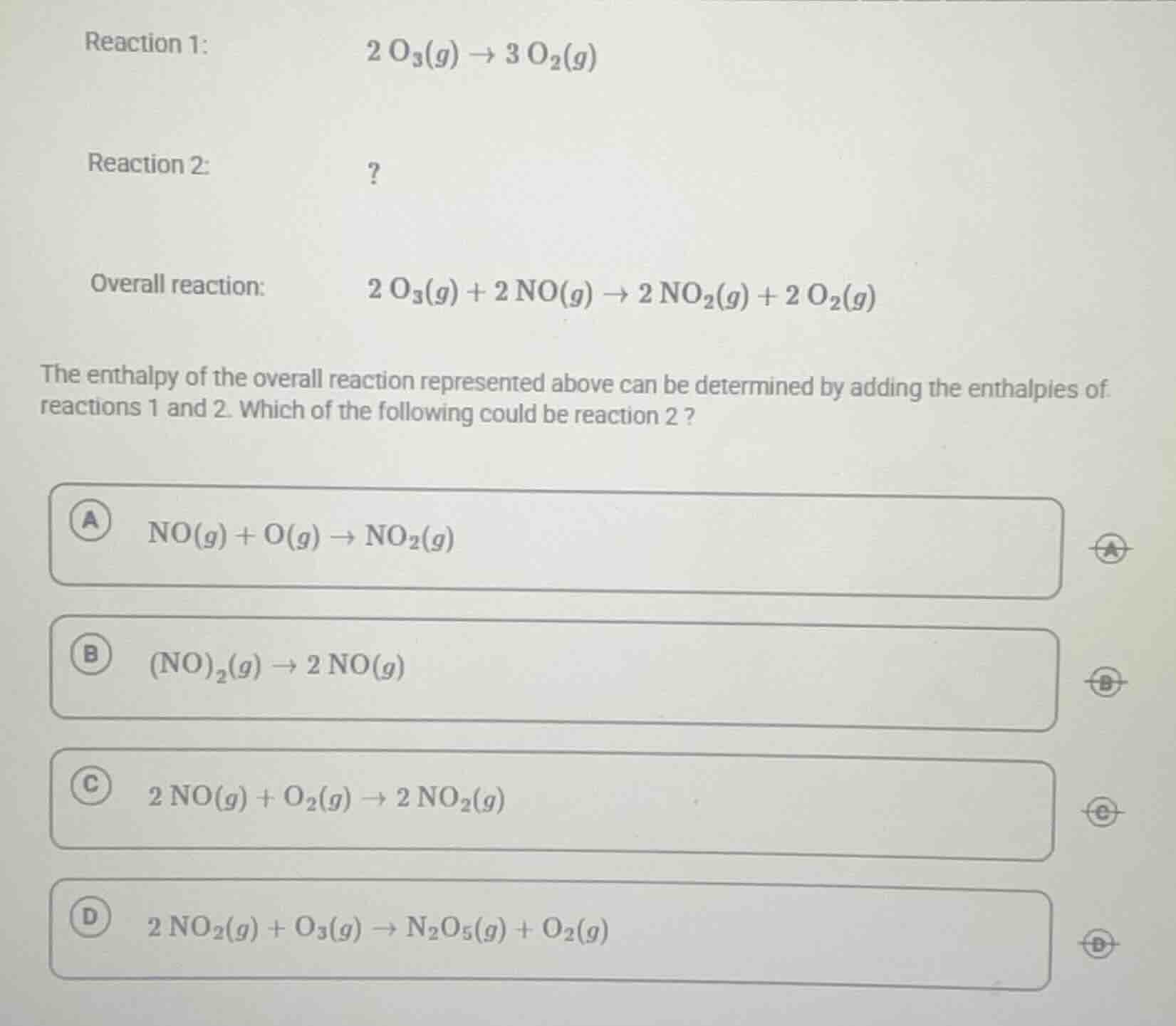
reaction 1: \\( 2 \\, \text{o}_3(g) \
ightarrow 3 \\, \text{o}_2(g) \\)
reaction 2: ?
overall reaction: \\( 2 \\, \text{o}_3(g) + 2 \\, \text{no}(g) \
ightarrow 2 \\, \text{no}_2(g) + 2 \\, \text{o}_2(g) \\)
the enthalpy of the overall reaction represented above can be determined by adding the enthalpies of reactions 1 and 2. which of the following could be reaction 2 ?
a \\( \text{no}(g) + \text{o}(g) \
ightarrow \text{no}_2(g) \\)
b \\( (\text{no})_2(g) \
ightarrow 2 \\, \text{no}(g) \\)
c \\( 2 \\, \text{no}(g) + \text{o}_2(g) \
ightarrow 2 \\, \text{no}_2(g) \\)
d \\( 2 \\, \text{no}_2(g) + \text{o}_3(g) \
ightarrow \text{n}_2\text{o}_5(g) + \text{o}_2(g) \\)
Step1: Recall Hess's Law
Hess's Law states that the enthalpy of an overall reaction is the sum of the enthalpies of the individual reactions that add up to the overall reaction. So, we need to find a reaction (Reaction 2) such that when we add Reaction 1 and Reaction 2, we get the overall reaction.
Reaction 1: \( 2 \text{O}_3(g)
ightarrow 3 \text{O}_2(g) \)
Overall reaction: \( 2 \text{O}_3(g) + 2 \text{NO}(g)
ightarrow 2 \text{NO}_2(g) + 2 \text{O}_2(g) \)
Let's denote Reaction 2 as \( a \text{NO}(g) + b \text{O}_2(g) + \dots
ightarrow c \text{NO}_2(g) + \dots \) (we need to find the correct stoichiometry).
Step2: Subtract Reaction 1 from the Overall Reaction
To find Reaction 2, we can perform the operation: Overall Reaction - Reaction 1.
First, write the overall reaction and Reaction 1:
Overall: \( 2 \text{O}_3(g) + 2 \text{NO}(g)
ightarrow 2 \text{NO}_2(g) + 2 \text{O}_2(g) \)
Reaction 1: \( 2 \text{O}_3(g)
ightarrow 3 \text{O}_2(g) \)
Subtracting Reaction 1 from the overall reaction (which is equivalent to adding the reverse of Reaction 1, but here we just subtract the species):
Left side: \( (2 \text{O}_3 + 2 \text{NO}) - (2 \text{O}_3) = 2 \text{NO} \)
Right side: \( (2 \text{NO}_2 + 2 \text{O}_2) - (3 \text{O}_2) = 2 \text{NO}_2 - \text{O}_2 \) Wait, no, actually, when subtracting reactions, we can think of it as:
Overall reaction = Reaction 1 + Reaction 2
So, Reaction 2 = Overall reaction - Reaction 1
Let's do the species by species:
For \( \text{O}_3 \): 2 in overall, 2 in Reaction 1, so 2 - 2 = 0 (so \( \text{O}_3 \) cancels out)
For \( \text{NO} \): 2 in overall, 0 in Reaction 1, so 2 in Reaction 2 (reactant)
For \( \text{O}_2 \): 2 in overall, 3 in Reaction 1 (but Reaction 1 has \( \text{O}_2 \) as product, so when we subtract Reaction 1, we reverse it? Wait, no, let's use the method of adding reactions.
Let me write Reaction 2 as \( x \text{NO}(g) + y \text{O}_2(g)
ightarrow z \text{NO}_2(g) \) (we need to find x, y, z)
Adding Reaction 1 and Reaction 2:
Reaction 1: \( 2 \text{O}_3
ightarrow 3 \text{O}_2 \)
Reaction 2: \( x \text{NO} + y \text{O}_2
ightarrow z \text{NO}_2 \)
Sum: \( 2 \text{O}_3 + x \text{NO} + y \text{O}_2
ightarrow 3 \text{O}_2 + z \text{NO}_2 \)
This should equal the overall reaction: \( 2 \text{O}_3 + 2 \text{NO}
ightarrow 2 \text{NO}_2 + 2 \text{O}_2 \)
Now, equate the coefficients:
For \( \text{NO} \): x = 2
For \( \text{NO}_2 \): z = 2
For \( \text{O}_2 \): y + 3 (from Reaction 1's product) = 2 (from overall's product) +? Wait, no, in the sum, the right side is \( 3 \text{O}_2 + z \text{NO}_2 \), and the overall right side is \( 2 \text{NO}_2 + 2 \text{O}_2 \). Wait, I think I messed up the direction. Let's instead write Reaction 2 as a reaction that, when added to Reaction 1, gives the overall reaction.
So:
Reaction 1: \( 2 \text{O}_3
ightarrow 3 \text{O}_2 \)
Reaction 2:?
Sum: \( 2 \text{O}_3 + 2 \text{NO}
ightarrow 2 \text{NO}_2 + 2 \text{O}_2 \)
Let's rearrange:
Reaction 2 = Sum - Reaction 1
So, (Sum) - (Reaction 1) is:
\( (2 \text{O}_3 + 2 \text{NO}
ightarrow 2 \text{NO}_2 + 2 \text{O}_2) - (2 \text{O}_3
ightarrow 3 \text{O}_2) \)
Which is equivalent to:
\( 2 \text{O}_3 + 2 \text{NO} - 2 \text{O}_3
ightarrow 2 \text{NO}_2 + 2 \text{O}_2 - 3 \text{O}_2 \)
Simplify:
\( 2 \text{NO}
ightarrow 2 \text{NO}_2 - \text{O}_2 \)
Wait, that can't be. Wait, no, when subtracting reactions, we can think of it as adding the reverse of Reaction 1. The reverse of Reaction 1 is \( 3 \text{O}_2
ightarrow 2 \text{O}_3 \)
Now, add reverse of Reaction 1 to the overall re…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. \( 2 \text{NO}(g) + \text{O}_2(g)
ightarrow 2 \text{NO}_2(g) \)