QUESTION IMAGE
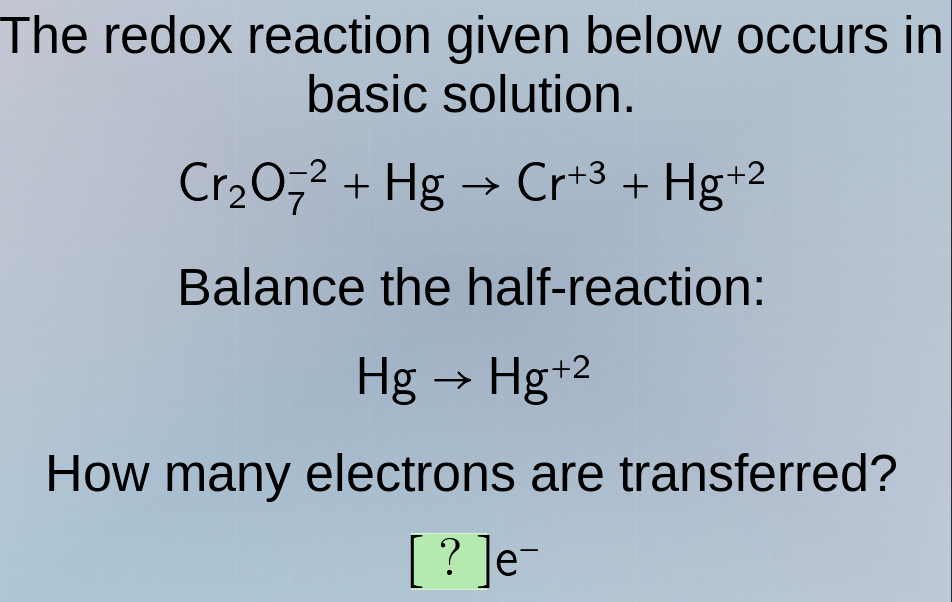
Question
the redox reaction given below occurs in basic solution.\\(\ce{cr2o7^{-2} + hg -> cr^{+3} + hg^{+2}}\\)\
balance the half-reaction:\\(\ce{hg -> hg^{+2}}\\)\
how many electrons are transferred?\\(\ce{?e^-}\\)
Step1: Determine oxidation state change
Hg has an oxidation state of 0 in elemental form. In \(Hg^{2+}\), the oxidation state is +2. So the change in oxidation state is \(+2 - 0 = +2\).
Step2: Relate to electron transfer
For oxidation (loss of electrons), the number of electrons lost is equal to the change in oxidation state. Since Hg goes from 0 to +2, it loses 2 electrons. The half - reaction is \(Hg
ightarrow Hg^{2 +}+2e^{-}\), so 2 electrons are transferred.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2