QUESTION IMAGE
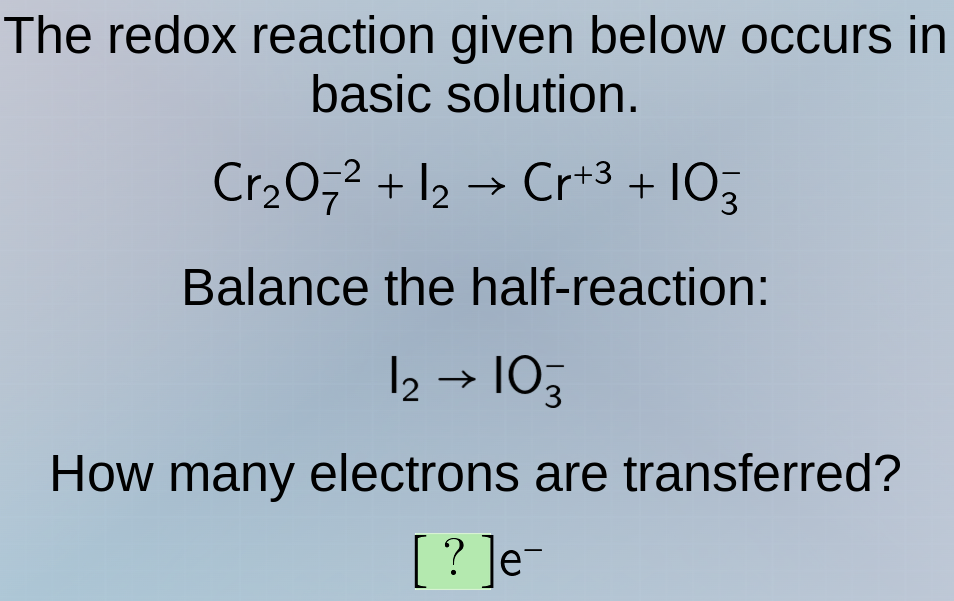
Question
the redox reaction given below occurs in basic solution.
$\ce{cr_{2}o^{ - 2}_{7} + i_{2} \
ightarrow cr^{ + 3} + io^{ - }_{3}}$
balance the half - reaction:
$\ce{i_{2} \
ightarrow io^{ - }_{3}}$
how many electrons are transferred?
$?\ce{e^{ - }}$
Step1: Balance atoms other than O and H
Start with the given half - reaction \(I_2
ightarrow IO_3^-\). First, balance the iodine atoms. There are 2 I atoms on the left and 1 I atom on the right. So we multiply \(IO_3^-\) by 2:
\(I_2
ightarrow 2IO_3^-\)
Step2: Balance oxygen atoms by adding \(H_2O\)
On the right - hand side, we have \(2\times3 = 6\) O atoms. So we add 6 \(H_2O\) molecules to the left - hand side to balance the O atoms:
\(I_2+6H_2O
ightarrow 2IO_3^-\)
Step3: Balance hydrogen atoms by adding \(H^+\)
On the left - hand side, we have \(6\times2=12\) H atoms from \(H_2O\). So we add 12 \(H^+\) ions to the right - hand side:
\(I_2 + 6H_2O
ightarrow 2IO_3^-+12H^+\)
Step4: Since the reaction is in basic solution, add \(OH^-\) to both sides to neutralize \(H^+\)
Add 12 \(OH^-\) to both sides:
\(I_2+6H_2O + 12OH^-
ightarrow 2IO_3^-+12H^++12OH^-\)
The \(12H^+\) and \(12OH^-\) on the right - hand side combine to form \(12H_2O\):
\(I_2+6H_2O + 12OH^-
ightarrow 2IO_3^-+12H_2O\)
Simplify the \(H_2O\) molecules. Subtract 6 \(H_2O\) from both sides:
\(I_2+12OH^-
ightarrow 2IO_3^-+6H_2O\)
Step5: Balance the charge by adding electrons
First, calculate the total charge on each side.
Left - hand side: Charge of \(I_2\) is 0, charge of \(12OH^-\) is \(- 12\), so total charge is \(-12\).
Right - hand side: Charge of \(2IO_3^-\) is \(2\times(-1)=-2\), charge of \(6H_2O\) is 0, so total charge is \(-2\).
Let the number of electrons be \(x\). Electrons have a charge of \(-1\) each. We need to balance the charge:
\(-12 + x=-2\)
Solving for \(x\), we get \(x = 10\). So we add 10 electrons to the right - hand side:
\(I_2+12OH^-
ightarrow 2IO_3^-+6H_2O + 10e^-\)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
10