QUESTION IMAGE
Question
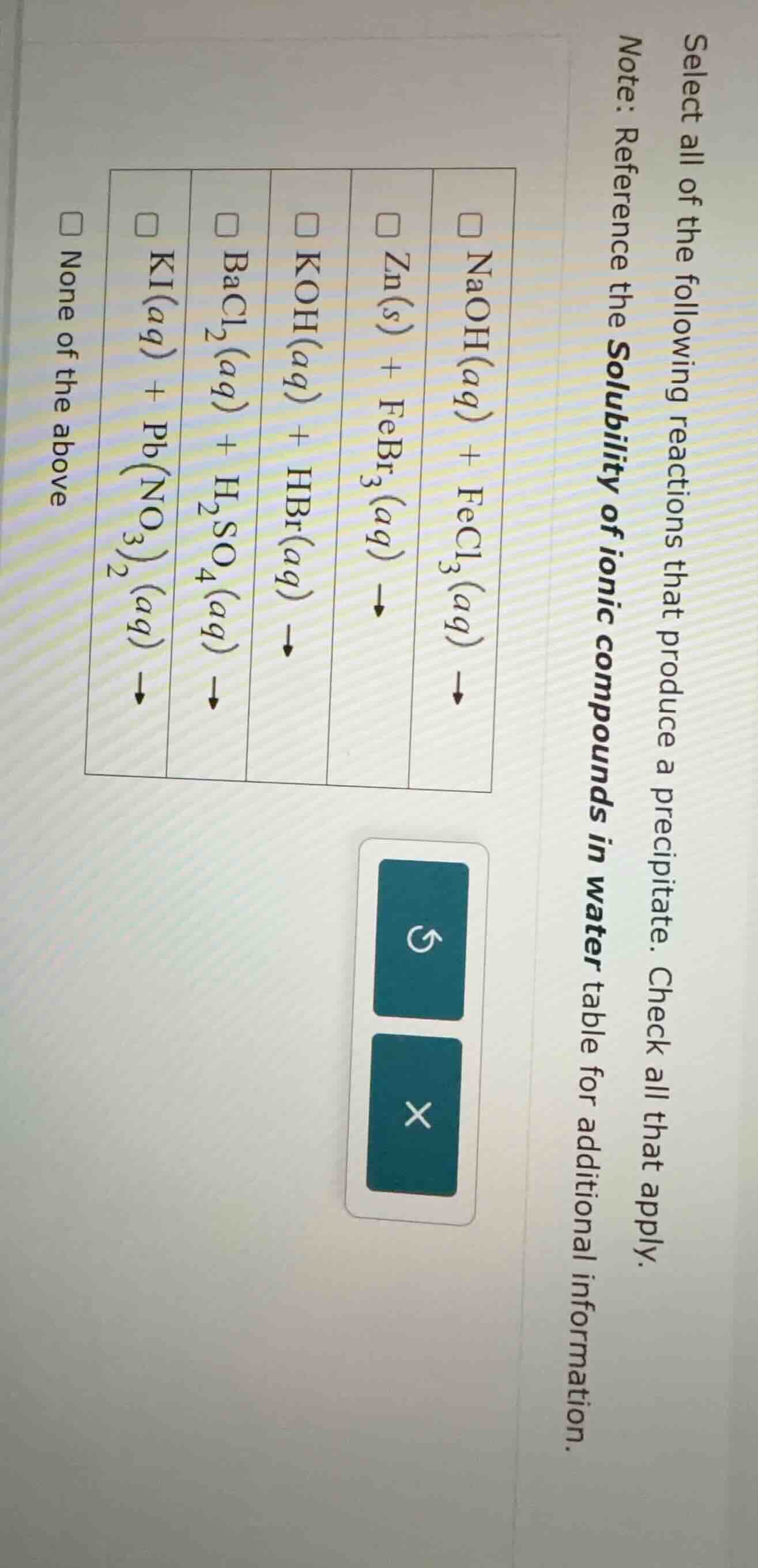
select all of the following reactions that produce a precipitate. check all that apply. note: reference the solubility of ionic compounds in water table for additional information. □ naoh(aq) + fecl₃(aq) → □ zn(s) + febr₃(aq) → □ koh(aq) + hbr(aq) → □ bacl₂(aq) + h₂so₄(aq) → □ ki(aq) + pb(no₃)₂(aq) → □ none of the above
Brief Explanations
- Analyze $\ce{NaOH(aq) + FeCl_{3}(aq)}$: The reaction is $\ce{3NaOH(aq) + FeCl_{3}(aq) = Fe(OH)_{3}(s) + 3NaCl(aq)}$. $\ce{Fe(OH)_{3}}$ is insoluble (precipitate).
- Analyze $\ce{Zn(s) + FeBr_{3}(aq)}$: This is a single - replacement reaction, $\ce{3Zn(s) + 2FeBr_{3}(aq) = 3ZnBr_{2}(aq) + 2Fe(s)}$. The product $\ce{Fe(s)}$ is a solid (precipitate - like, as it's a metal formed from solution).
- Analyze $\ce{KOH(aq) + HBr(aq)}$: The reaction is $\ce{KOH(aq) + HBr(aq) = KBr(aq) + H_{2}O(l)}$. No precipitate (both $\ce{KBr}$ and $\ce{H_{2}O}$ are soluble/liquid).
- Analyze $\ce{BaCl_{2}(aq) + H_{2}SO_{4}(aq)}$: The reaction is $\ce{BaCl_{2}(aq) + H_{2}SO_{4}(aq) = BaSO_{4}(s) + 2HCl(aq)}$. $\ce{BaSO_{4}}$ is insoluble (precipitate).
- Analyze $\ce{KI(aq) + Pb(NO_{3})_{2}(aq)}$: The reaction is $\ce{2KI(aq) + Pb(NO_{3})_{2}(aq) = PbI_{2}(s) + 2KNO_{3}(aq)}$. $\ce{PbI_{2}}$ is insoluble (precipitate).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\ce{NaOH(aq) + FeCl_{3}(aq) ->}$ (produces $\ce{Fe(OH)_{3}}$ precipitate)
- $\ce{Zn(s) + FeBr_{3}(aq) ->}$ (produces $\ce{Fe}$ precipitate)
- $\ce{BaCl_{2}(aq) + H_{2}SO_{4}(aq) ->}$ (produces $\ce{BaSO_{4}}$ precipitate)
- $\ce{KI(aq) + Pb(NO_{3})_{2}(aq) ->}$ (produces $\ce{PbI_{2}}$ precipitate)