QUESTION IMAGE
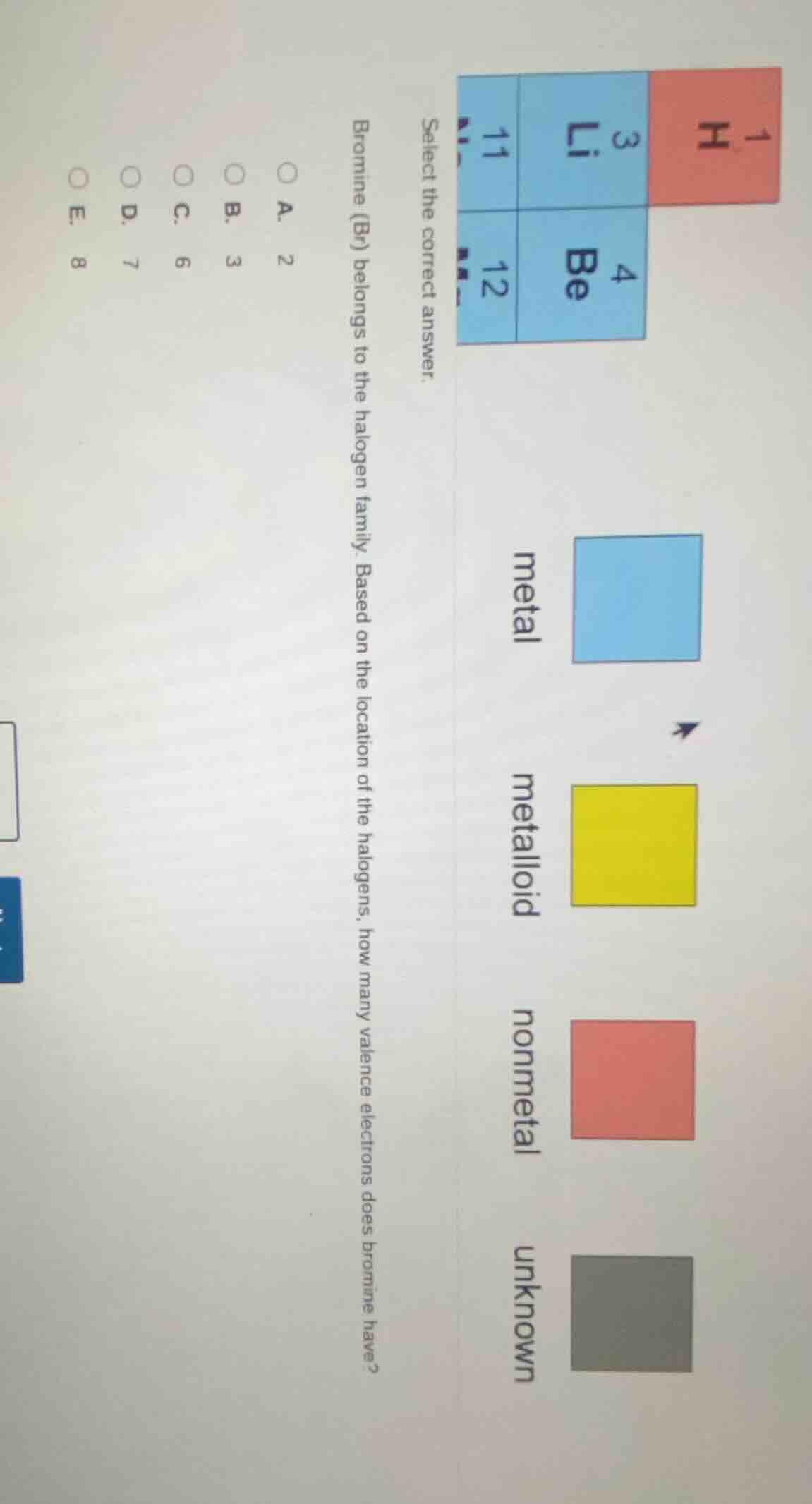
Question
select the correct answer.
bromine (br) belongs to the halogen family. based on the location of the halogens, how many valence electrons does bromine have?
a. 2
b. 3
c. 6
d. 7
e. 8
Step1: Identify halogen group
Halogens are in Group 17 (or 7A) of the periodic table. Bromine (Br) is a halogen, so it belongs to Group 17.
Step2: Determine valence electrons
Elements in Group 17 have 7 valence electrons (since the group number (17) minus 10 for main - group elements gives the number of valence electrons: \(17 - 10=7\)).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. 7