QUESTION IMAGE
Question
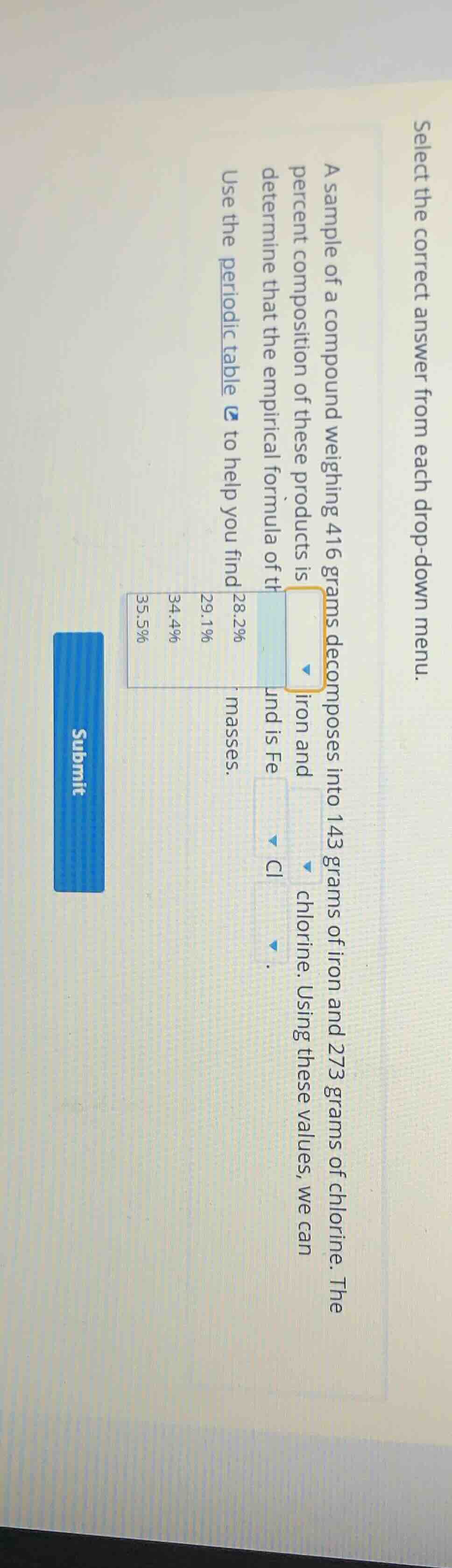
select the correct answer from each drop - down menu. a sample of a compound weighing 416 grams decomposes into 143 grams of iron and 273 grams of chlorine. the percent composition of these products is drop - down, iron and drop - down, chlorine. using these values, we can determine that the empirical formula of the compound is fe drop - down cl drop - down. use the periodic table to help you find atomic masses. chart with 28.2%, 29.1%, 34.4%, 35.5% submit
Step1: Calculate moles of Fe
Molar mass of Fe is 55.85 g/mol. Moles of Fe = $\frac{143\ g}{55.85\ g/mol}$ ≈ 2.56 mol.
Step2: Calculate moles of Cl
Molar mass of Cl is 35.45 g/mol. Moles of Cl = $\frac{273\ g}{35.45\ g/mol}$ ≈ 7.70 mol.
Step3: Find mole ratio
Divide moles by the smallest (2.56). Ratio of Fe: $\frac{2.56}{2.56}$ = 1. Ratio of Cl: $\frac{7.70}{2.56}$ ≈ 3. So formula is FeCl₃.
Step4: Calculate percent composition of Fe
Percent of Fe = $\frac{143}{416}×100$ ≈ 34.4%.
Step5: Calculate percent composition of Cl
Percent of Cl = $\frac{273}{416}×100$ ≈ 65.6%? Wait, no, wait the options: Wait the first drop - down (percent of Fe? Wait the problem says "the percent composition of these products is [drop - down] iron and [drop - down] chlorine". Wait the sample is 416g, Fe is 143g, Cl is 273g.
Percent of Fe: $\frac{143}{416}×100$ ≈ 34.4% (matches one of the options: 34.4%).
Percent of Cl: $\frac{273}{416}×100$ ≈ 65.6%? Wait no, the options given are 28.2%, 29.1%, 34.4%, 35.5%. Wait maybe I misread. Wait the problem says "the percent composition of these products is [ ] iron and [ ] chlorine. Using these values, we can determine that the empirical formula of the compound is Fe [ ] Cl [ ]." Wait maybe the first drop - down is percent of Fe: 143/416*100 = 34.4%, so that's the first. Then for the formula, FeCl₃, so the subscripts: Fe is 1, Cl is 3? Wait no, let's recalculate moles again.
Wait 143g Fe: 143 / 55.85 ≈ 2.56 mol. 273g Cl: 273 / 35.45 ≈ 7.70 mol. 7.70 / 2.56 ≈ 3. So FeCl₃. So the empirical formula is FeCl₃. So the percent of Fe is 34.4%, which is one of the options.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
For the percent composition of iron: 34.4% (the option with 34.4%). For the empirical formula, the subscript of Fe is 1 and Cl is 3 (but based on the drop - down, if the first drop - down is percent of Fe, then 34.4%, and for the formula, FeCl₃, so Fe with subscript 1, Cl with subscript 3. But since the problem has drop - downs, the first percent (iron) is 34.4%, and the formula is FeCl₃ (so Fe₁Cl₃, but maybe the drop - down for Fe is 1 and Cl is 3, or the percent of Cl? Wait no, the first drop - down is for iron's percent: 143/416*100 = 34.4%, so that's the answer for the first drop - down.