QUESTION IMAGE
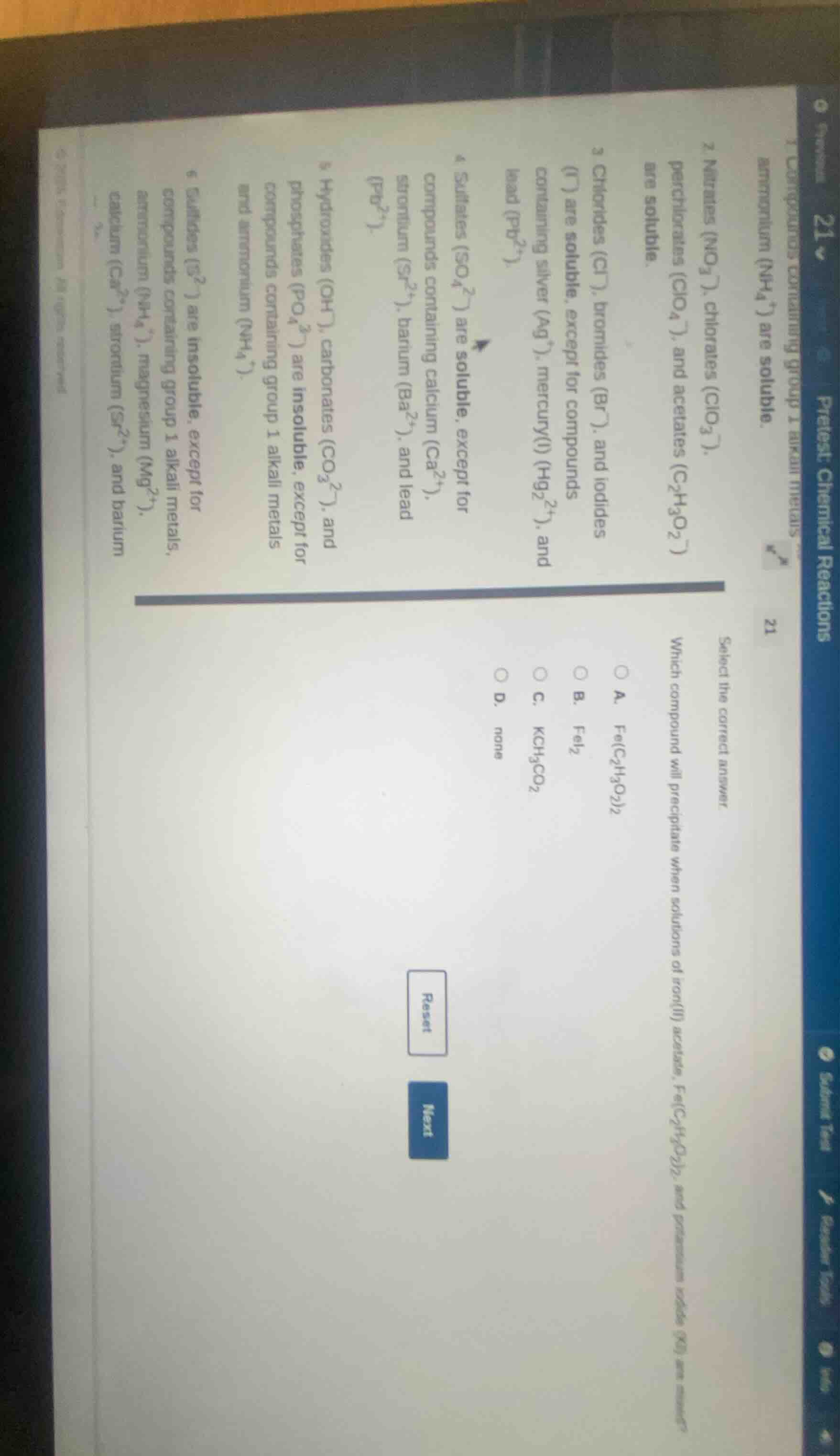
Question
select the correct answer
which compound will precipitate when solutions of iron(ii) acetate, fe(c₂h₃o₂)₂, and potassium iodide (ki) are mixed?
a. fe(c₂h₃o₂)₂
b. fei₂
c. kch₃co₂
d. none
To solve this, we analyze each option using solubility rules:
Step 1: Analyze Option A (\( \text{Fe}(\text{C}_2\text{H}_3\text{O}_2)_2 \))
Iron(II) acetate is one of the reactants. No precipitation occurs here (it’s the starting compound).
Step 2: Analyze Option B (\( \text{FeI}_2 \))
From solubility rule 3: Chlorides, bromides, iodides (\( \text{I}^- \)) are soluble, except for \( \text{Ag}^+ \), \( \text{Hg}_2^{2+} \), \( \text{Pb}^{2+} \). Iron(II) (\( \text{Fe}^{2+} \)) is not in the “insoluble” exception list for iodides. Thus, \( \text{FeI}_2 \) is soluble.
Step 3: Analyze Option C (\( \text{KCH}_3\text{CO}_2 \))
From solubility rule 2: Acetates (\( \text{C}_2\text{H}_3\text{O}_2^- \)) are soluble. Potassium (\( \text{K}^+ \)) is a Group 1 metal (rule 1: Group 1 compounds are soluble). Thus, potassium acetate is soluble.
Step 4: Analyze Option D (None)
Since \( \text{FeI}_2 \), \( \text{KCH}_3\text{CO}_2 \), and \( \text{Fe}(\text{C}_2\text{H}_3\text{O}_2)_2 \) are all soluble, no precipitate forms.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. none