QUESTION IMAGE
Question
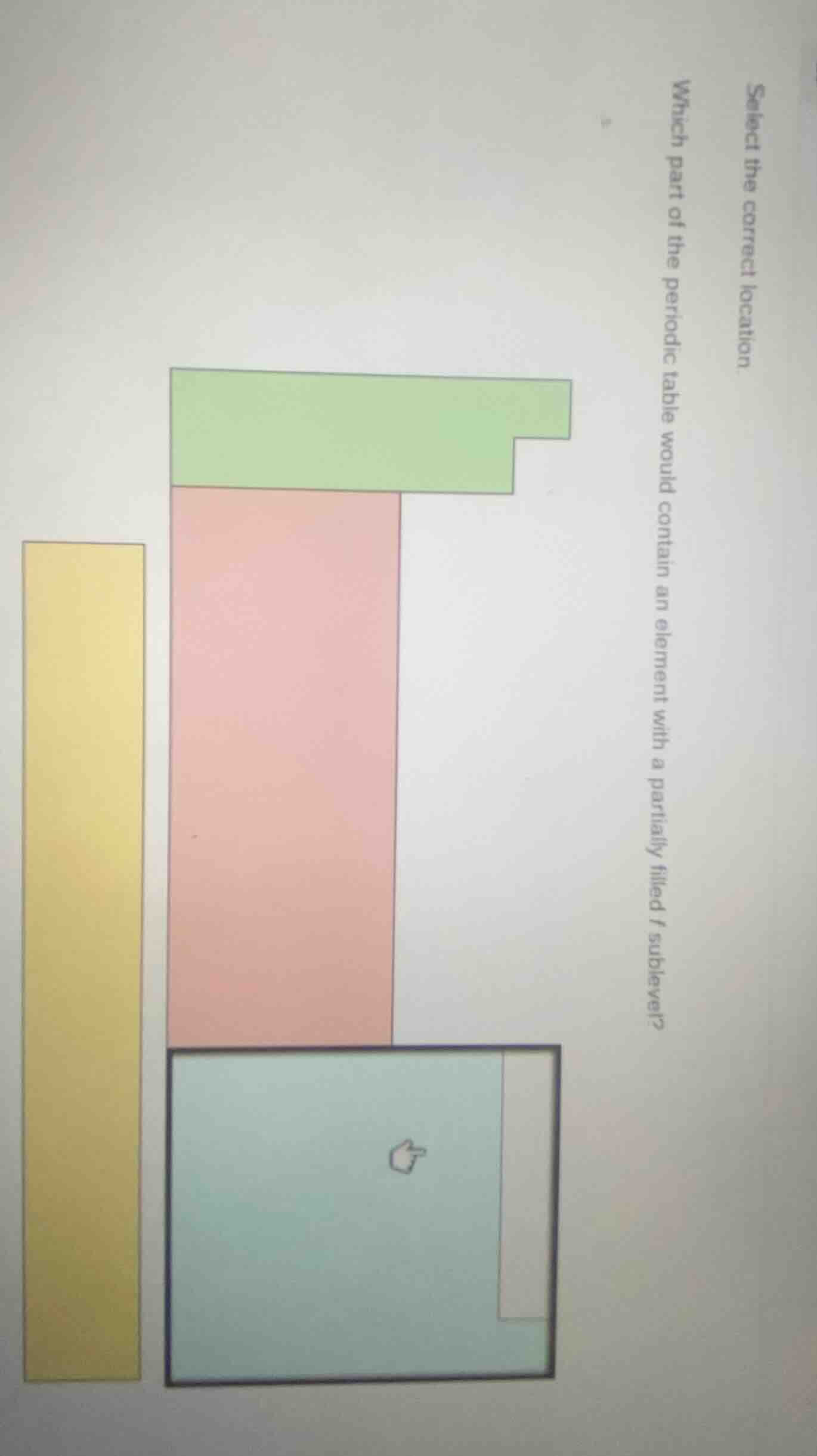
select the correct location. which part of the periodic table would contain an element with a partially filled f sublevel?
Elements with partially filled \( f \) sublevels are the lanthanides (filling 4\( f \)) and actinides (filling 5\( f \)), which are located in the bottom - block (the light - blue colored block in the diagram, often called the inner transition metals) of the periodic table. The green block is typically the main - group elements (s - and p - block, no \( f \) - sublevel filling), the pink block is the d - block (transition metals, filling d - sublevels), and the yellow block is also not the \( f \) - block. So the part with the light - blue color (the bottom block with the cursor) contains elements with partially filled \( f \) sublevels.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The light - blue colored block (the bottom block in the diagram, representing inner transition metals: lanthanides and actinides)