QUESTION IMAGE
Question
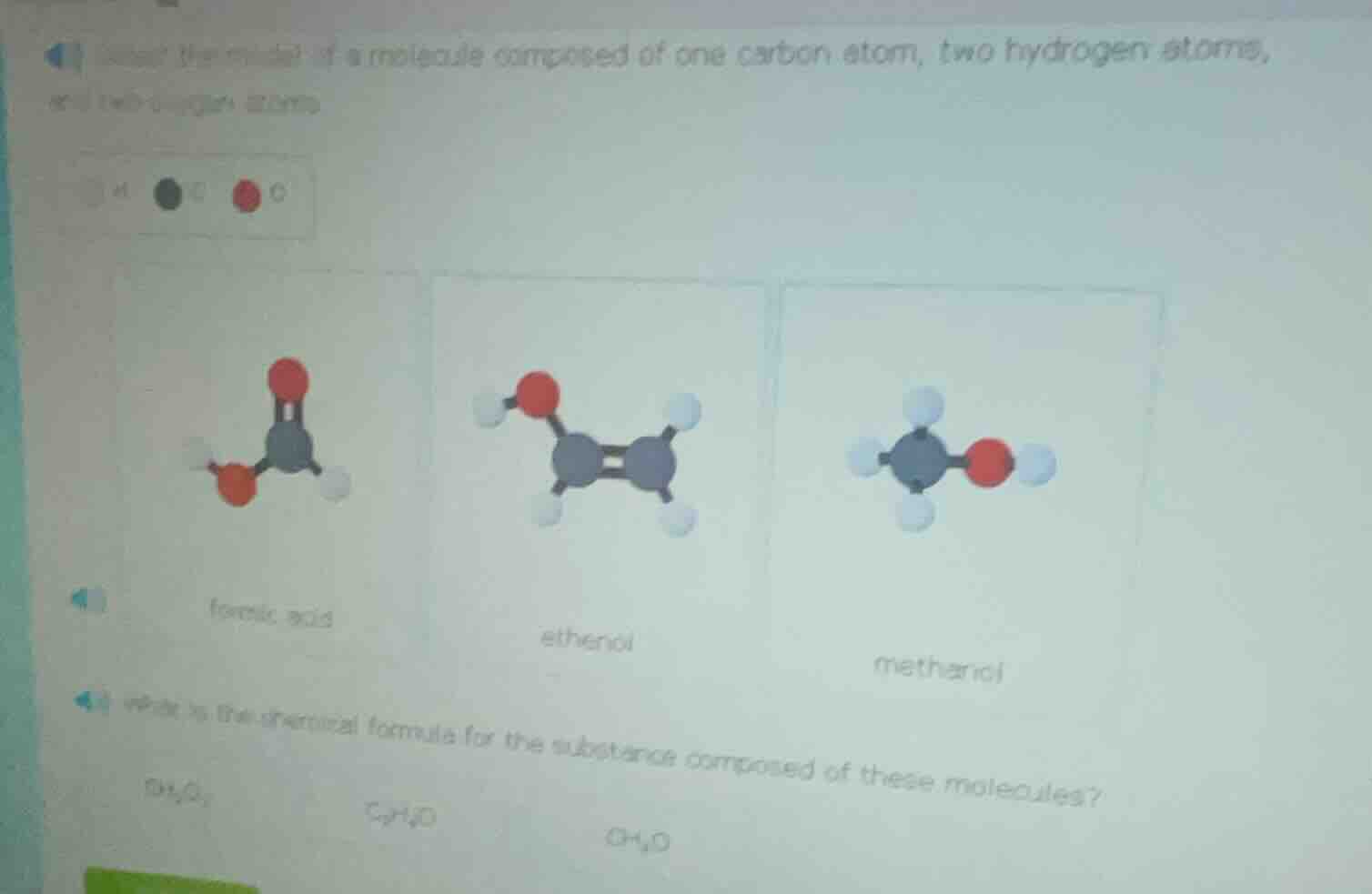
select the model of a molecule composed of one carbon atom, two hydrogen atoms, and two oxygen atoms
formic acid ethanol methanol
which is the chemical formula for the substance composed of these molecules?
c₃h₆o₂ c₂h₆o ch₂o
Part 1: Selecting the correct molecular model
The molecule has 1 C, 2 H, and 2 O atoms. Let’s analyze each model:
- Formic acid: Contains 1 C, 2 O, and 2 H (matches atom counts: C=1, H=2, O=2).
- Ethanol: Has 2 C atoms (too many, we need 1 C).
- Methanol: Has 1 C, 1 O, and 4 H (O count is 1, not 2).
Thus, the formic acid model (first one) is correct.
Step 1: Identify atom counts
The molecule has 1 carbon (C), 2 hydrogen (H), and 2 oxygen (O) atoms.
Step 2: Write the formula
Using the atom counts, the formula is $CH_2O_2$ (since C=1, H=2, O=2; subscripts are omitted for 1, so $CH_2O_2$).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The first model (labeled "formic acid")