QUESTION IMAGE
Question
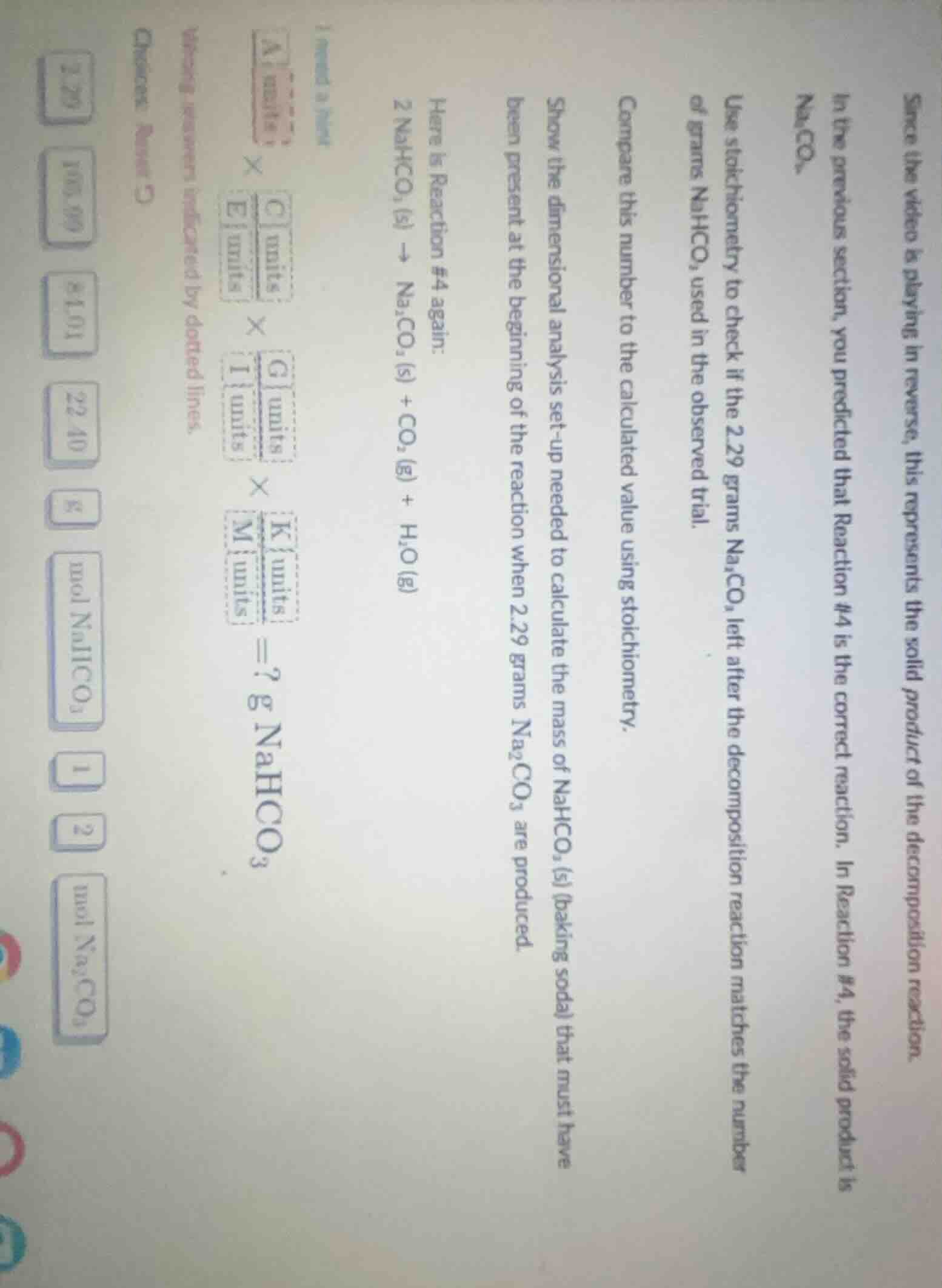
since the video is playing in reverse, this represents the solid product of the decomposition reaction.
in the previous section, you predicted that reaction #4 is the correct reaction. in reaction #4, the solid product is $ce{na_{2}co_{3}}$.
use stoichiometry to check if the 2.29 grams $ce{na_{2}co_{3}}$ left after the decomposition reaction matches the number of grams $ce{nahco_{3}}$ used in the observed trial.
compare this number to the calculated value using stoichiometry.
show the dimensional analysis set-up needed to calculate the mass of $ce{nahco_{3}}$(s) (baking soda) that must have been present at the beginning of the reaction when 2.29 grams $ce{na_{2}co_{3}}$ are produced.
here is reaction #4 again:
$ce{2 nahco_{3} (s) na_{2}co_{3} (s) + co_{2} (g) + h_{2}o (g)}$
$\frac{\text{a units}}{\text{ }} \times \frac{\text{c units}}{\text{e units}} \times \frac{\text{g units}}{\text{i units}} \times \frac{\text{k units}}{\text{m units}} = ? \text{ g } ce{nahco_{3}}$
wrong answers indicated by dotted lines.
choices: reset c
2.29 105.99 84.01 22.40 g mol $ce{nahco_{3}}$ 1 2 mol $ce{na_{2}co_{3}}$
Step1: Start with given mass
$2.29\ \text{g}\ \text{Na}_2\text{CO}_3$
Step2: Convert to moles of $\text{Na}_2\text{CO}_3$
$\text{Molar mass of }\text{Na}_2\text{CO}_3 = 2\times23 + 12 + 3\times16 = 105.99\ \text{g/mol}$
$2.29\ \text{g}\ \text{Na}_2\text{CO}_3 \times \frac{1\ \text{mol}\ \text{Na}_2\text{CO}_3}{105.99\ \text{g}\ \text{Na}_2\text{CO}_3}$
Step3: Mole ratio from reaction
From $2\text{NaHCO}_3(s)
ightarrow \text{Na}_2\text{CO}_3(s) + \text{CO}_2(g) + \text{H}_2\text{O}(g)$, mole ratio $\frac{2\ \text{mol}\ \text{NaHCO}_3}{1\ \text{mol}\ \text{Na}_2\text{CO}_3}$
$2.29\ \text{g}\ \text{Na}_2\text{CO}_3 \times \frac{1\ \text{mol}\ \text{Na}_2\text{CO}_3}{105.99\ \text{g}\ \text{Na}_2\text{CO}_3} \times \frac{2\ \text{mol}\ \text{NaHCO}_3}{1\ \text{mol}\ \text{Na}_2\text{CO}_3}$
Step4: Convert to grams of $\text{NaHCO}_3$
$\text{Molar mass of }\text{NaHCO}_3 = 23 + 1 + 12 + 3\times16 = 84.01\ \text{g/mol}$
$2.29\ \text{g}\ \text{Na}_2\text{CO}_3 \times \frac{1\ \text{mol}\ \text{Na}_2\text{CO}_3}{105.99\ \text{g}\ \text{Na}_2\text{CO}_3} \times \frac{2\ \text{mol}\ \text{NaHCO}_3}{1\ \text{mol}\ \text{Na}_2\text{CO}_3} \times \frac{84.01\ \text{g}\ \text{NaHCO}_3}{1\ \text{mol}\ \text{NaHCO}_3}$
Step5: Calculate final value
$\frac{2.29 \times 2 \times 84.01}{105.99} \approx 3.57\ \text{g}$
Filled dimensional analysis:
$2.29\ \text{g}\ \text{Na}_2\text{CO}_3 \times \frac{1\ \text{mol}\ \text{Na}_2\text{CO}_3}{105.99\ \text{g}\ \text{Na}_2\text{CO}_3} \times \frac{2\ \text{mol}\ \text{NaHCO}_3}{1\ \text{mol}\ \text{Na}_2\text{CO}_3} \times \frac{84.01\ \text{g}\ \text{NaHCO}_3}{1\ \text{mol}\ \text{NaHCO}_3} = 3.57\ \text{g}\ \text{NaHCO}_3$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The mass of $\text{NaHCO}_3$ required is approximately $\boldsymbol{3.57\ \text{g}}$, with the dimensional analysis filled as:
$2.29\ \text{g}\ \text{Na}_2\text{CO}_3 \times \frac{1\ \text{mol}\ \text{Na}_2\text{CO}_3}{105.99\ \text{g}\ \text{Na}_2\text{CO}_3} \times \frac{2\ \text{mol}\ \text{NaHCO}_3}{1\ \text{mol}\ \text{Na}_2\text{CO}_3} \times \frac{84.01\ \text{g}\ \text{NaHCO}_3}{1\ \text{mol}\ \text{NaHCO}_3}$