QUESTION IMAGE
Question
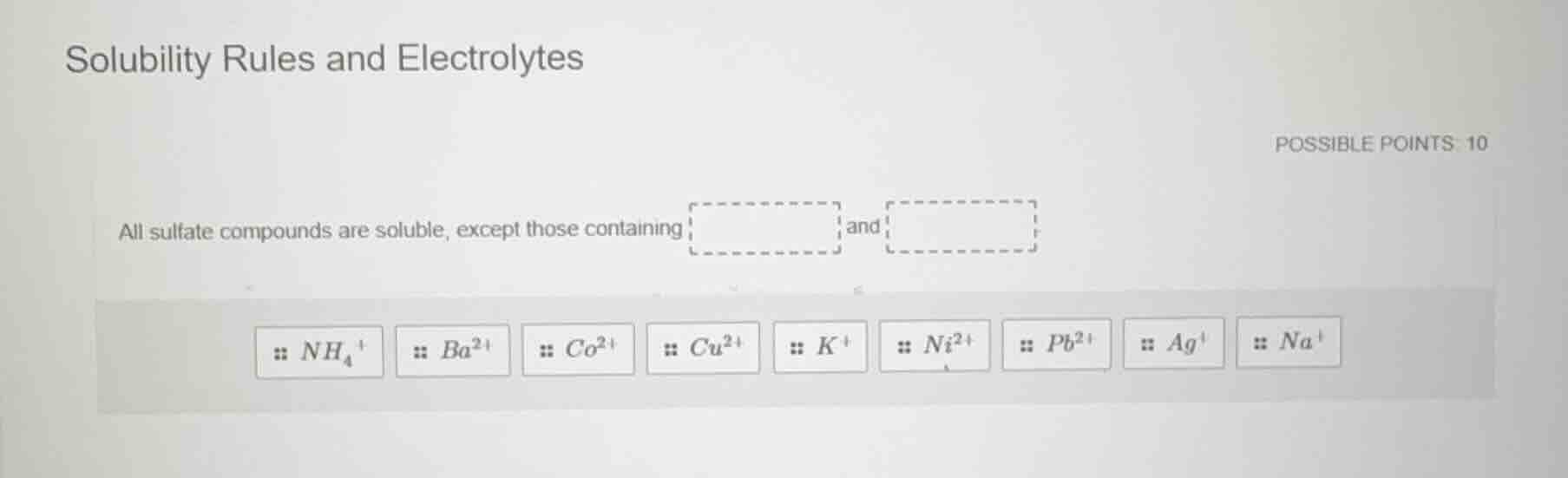
solubility rules and electrolytes
possible points: 10
all sulfate compounds are soluble, except those containing ____ and ____
$nh_4^+$ $ba^{2+}$ $co^{2+}$ $cu^{2+}$ $k^+$ $ni^{2+}$ $pb^{2+}$ $ag^+$ $na^+$
Brief Explanations
Solubility rules state that sulfate compounds ($SO_4^{2-}$) are generally soluble in water, but form insoluble (or sparingly soluble) compounds with specific cations. From the given options, these cations are barium and lead(II).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{Ba^{2+}}$ and $\boldsymbol{Pb^{2+}}$