QUESTION IMAGE
Question
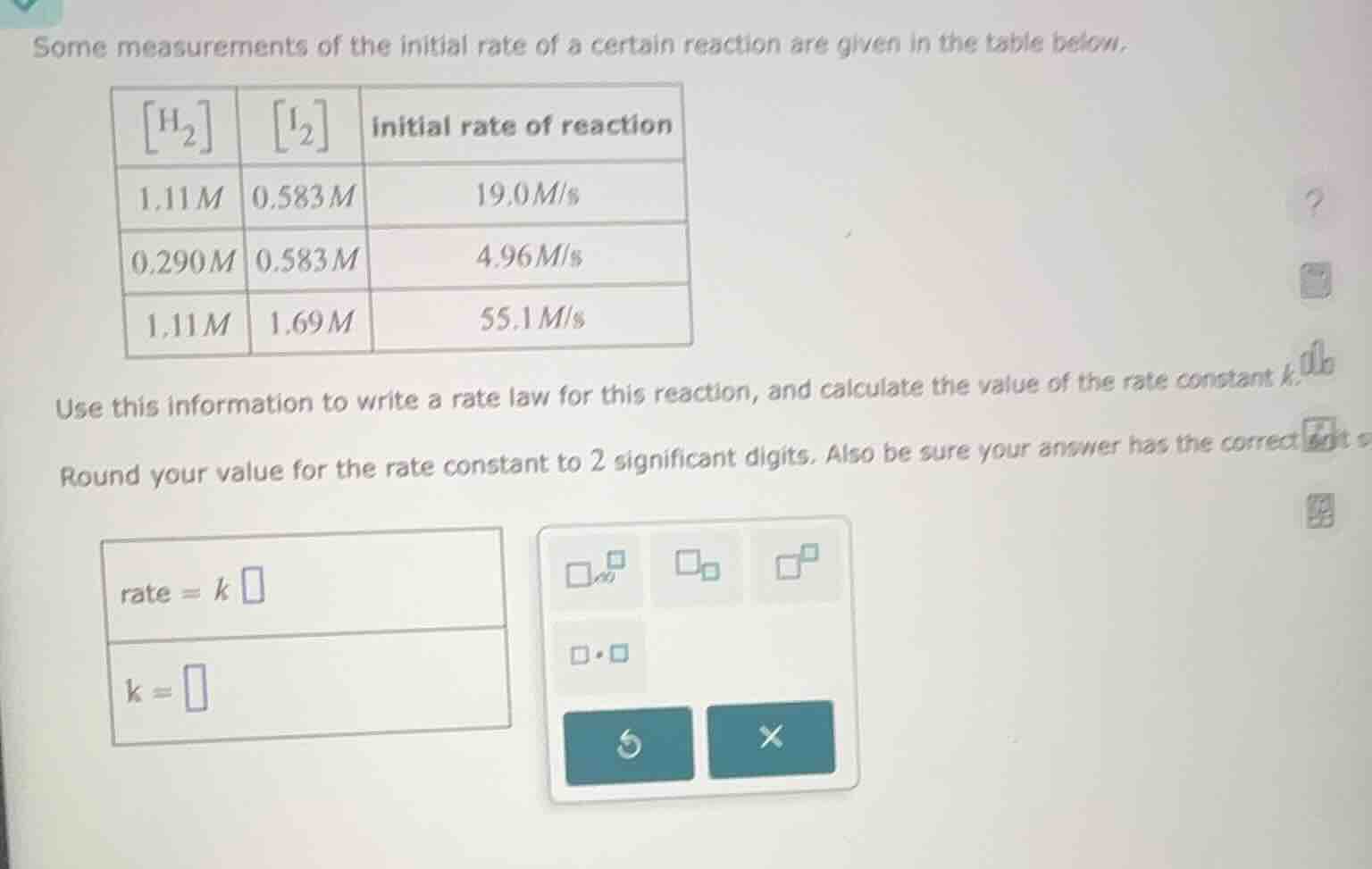
some measurements of the initial rate of a certain reaction are given in the table below.
| $h_2$ | $i_2$ | initial rate of reaction | ---- | ---- | ---- | $1.11m$ | $0.583m$ | $19.0m/s$ | $0.290m$ | $0.583m$ | $4.96m/s$ | $1.11m$ | $1.69m$ | $55.1m/s$ | use this information to write a rate law for this reaction, and calculate the value of the rate constant $k$.round your value for the rate constant to 2 significant digits. also be sure your answer has the correct$rate = k \square$$k = \square$ |
Step1: Define general rate law
The general rate law is $\text{rate} = k[\text{H}_2]^x[\text{I}_2]^y$, where $x$ and $y$ are reaction orders.
Step2: Solve for $x$ using rows 1&2
Divide rate1 by rate2 (hold $[\text{I}_2]$ constant):
$$\frac{19.0\ M/s}{4.96\ M/s} = \frac{k(1.11\ M)^x(0.583\ M)^y}{k(0.290\ M)^x(0.583\ M)^y}$$
Simplify: $3.83 = (3.83)^x$, so $x=1$.
Step3: Solve for $y$ using rows 1&3
Divide rate3 by rate1 (hold $[\text{H}_2]$ constant):
$$\frac{55.1\ M/s}{19.0\ M/s} = \frac{k(1.11\ M)^x(1.69\ M)^y}{k(1.11\ M)^x(0.583\ M)^y}$$
Simplify: $2.90 = (2.90)^y$, so $y=1$.
Step4: Write final rate law
Substitute $x=1$, $y=1$: $\text{rate} = k[\text{H}_2][\text{I}_2]$
Step5: Calculate $k$ with row1 data
Rearrange rate law: $k = \frac{\text{rate}}{[\text{H}_2][\text{I}_2]}$
Substitute values:
$$k = \frac{19.0\ M/s}{(1.11\ M)(0.583\ M)}$$
Calculate: $k = \frac{19.0}{0.64713}\ M^{-1}s^{-1} \approx 29.36\ M^{-1}s^{-1}$
Round to 2 sig figs: $k \approx 29\ M^{-1}s^{-1}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\text{rate} = k[\text{H}_2][\text{I}_2]$
$k = 29\ M^{-1}s^{-1}$