QUESTION IMAGE
Question
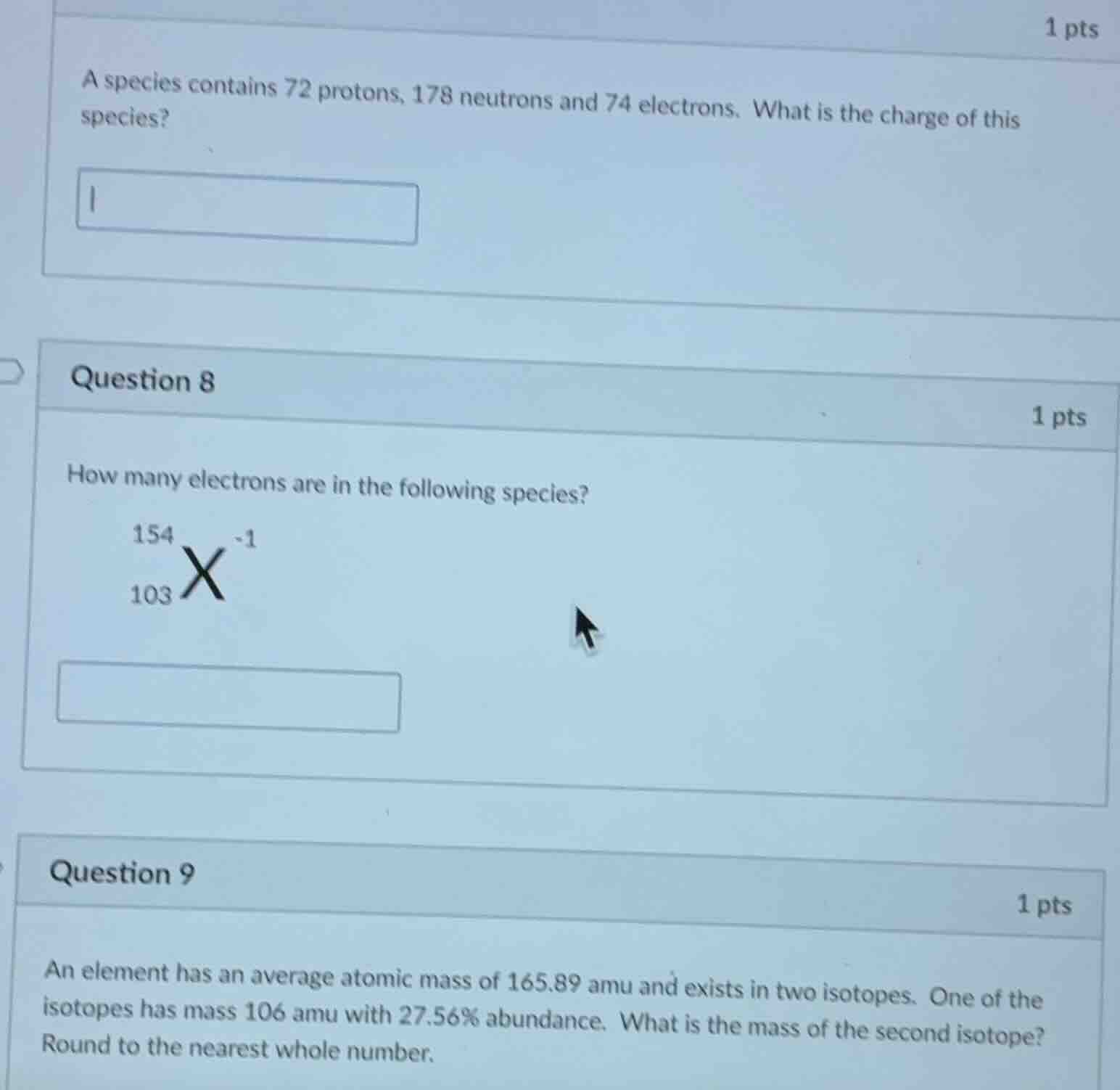
a species contains 72 protons, 178 neutrons and 74 electrons. what is the charge of this species?
question 8
how many electrons are in the following species?
\\(\ce{^{154}_{103}x^{-1}}\\)
question 9
an element has an average atomic mass of 165.89 amu and exists in two isotopes. one of the isotopes has mass 106 amu with 27.56% abundance. what is the mass of the second isotope? round to the nearest whole number.
Question (First one: Charge of the species)
Step1: Recall charge formula
The charge of a species is given by \( \text{Charge} = \text{Number of protons} - \text{Number of electrons} \) (since protons are positive and electrons are negative, neutrons have no charge).
Step2: Substitute values
Given, number of protons \( = 72 \), number of electrons \( = 74 \). So, \( \text{Charge} = 72 - 74 = -2 \).
Step1: Recall atomic number and charge relation
The atomic number (bottom number) is the number of protons, \( Z = 103 \). For a neutral atom, electrons = protons. But here, the charge is \(-1\), which means it has gained 1 electron.
Step2: Calculate electrons
So, number of electrons \( = \text{Number of protons} + \text{Magnitude of charge (since negative charge means extra electrons)} \). Thus, electrons \( = 103 + 1 = 104 \).
Step1: Recall average atomic mass formula
The average atomic mass (\( A_{\text{avg}} \)) is given by \( A_{\text{avg}} = f_1A_1 + f_2A_2 \), where \( f_1, f_2 \) are the fractional abundances and \( A_1, A_2 \) are the masses of the isotopes. Given \( f_1 = 0.2756 \) (27.56% converted to fraction), \( A_1 = 106 \) amu, \( A_{\text{avg}} = 165.89 \) amu, and \( f_2 = 1 - f_1 = 1 - 0.2756 = 0.7244 \).
Step2: Rearrange formula to solve for \( A_2 \)
From \( A_{\text{avg}} = f_1A_1 + f_2A_2 \), we get \( A_2=\frac{A_{\text{avg}} - f_1A_1}{f_2} \).
Substitute the values: \( A_2=\frac{165.89 - (0.2756\times106)}{0.7244} \).
First, calculate \( 0.2756\times106 = 29.2136 \). Then, \( 165.89 - 29.2136 = 136.6764 \). Now, divide by \( 0.7244 \): \( \frac{136.6764}{0.7244} \approx 189 \) (rounded to nearest whole number).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(-2\)