QUESTION IMAGE
Question
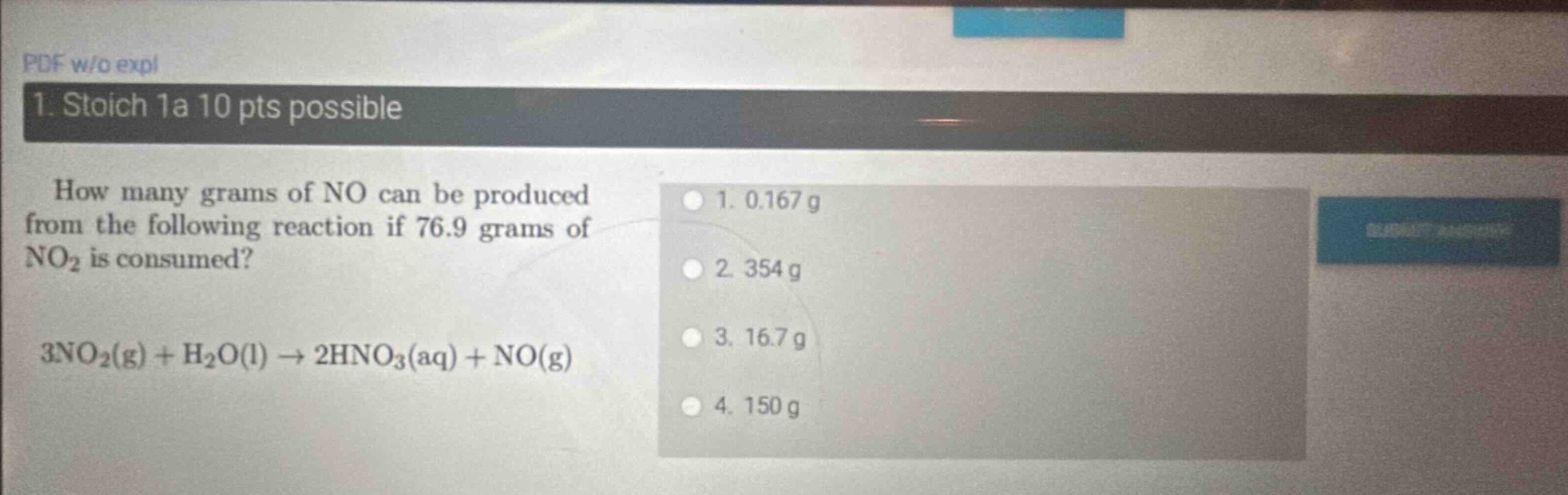
- stoich 1a 10 pts possible how many grams of no can be produced from the following reaction if 76.9 grams of no₂ is consumed? 3no₂(g) + h₂o(l) → 2hno₃(aq) + no(g) 1. 0.167 g 2. 354 g 3. 16.7 g 4. 150 g
Step1: Calculate moles of \( \text{NO}_2 \)
Molar mass of \( \text{NO}_2 \) is \( 14.01 + 2\times16.00 = 46.01 \, \text{g/mol} \). Moles of \( \text{NO}_2 = \frac{76.9 \, \text{g}}{46.01 \, \text{g/mol}} \approx 1.671 \, \text{mol} \).
Step2: Use stoichiometry to find moles of \( \text{NO} \)
From the reaction \( 3\text{NO}_2
ightarrow \text{NO} \), the mole ratio is \( \frac{1}{3} \). Moles of \( \text{NO} = \frac{1}{3} \times 1.671 \, \text{mol} \approx 0.557 \, \text{mol} \).
Step3: Calculate mass of \( \text{NO} \)
Molar mass of \( \text{NO} \) is \( 14.01 + 16.00 = 30.01 \, \text{g/mol} \). Mass of \( \text{NO} = 0.557 \, \text{mol} \times 30.01 \, \text{g/mol} \approx 16.7 \, \text{g} \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- 16.7 g