QUESTION IMAGE
Question
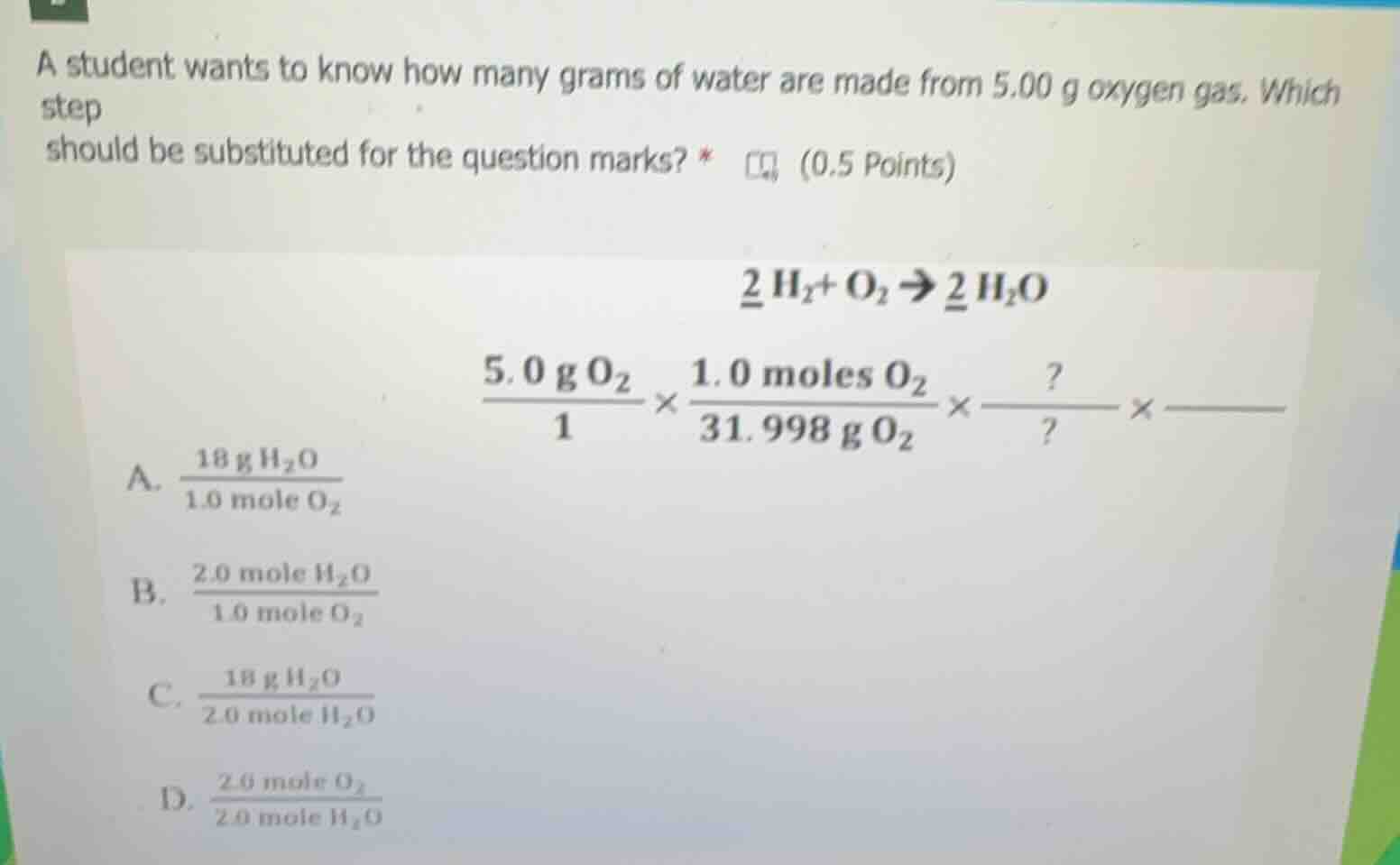
a student wants to know how many grams of water are made from 5.00 g oxygen gas. which step should be substituted for the question marks? * (0.5 points) \\(\underline{2}\ce{h2}+\ce{o2}\
ightarrow\underline{2}\ce{h2o}\\) \\(\frac{5.0\ce{g o2}}{1}\times\frac{1.0\ce{moles o2}}{31.998\ce{g o2}}\times\frac{?}{?}\times\dots\\) a. \\(\frac{18\ce{g h2o}}{1.0\ce{mole o2}}\\) b. \\(\frac{2.0\ce{mole h2o}}{1.0\ce{mole o2}}\\) c. \\(\frac{18\ce{g h2o}}{2.0\ce{mole h2o}}\\) d. \\(\frac{2.0\ce{mole o2}}{2.0\ce{mole h2o}}\\)
To solve for the mass of water from oxygen, we use stoichiometry. The balanced equation is \(2H_2 + O_2
ightarrow 2H_2O\), so the mole ratio of \(O_2\) to \(H_2O\) is \(1:2\) (1 mole \(O_2\) produces 2 moles \(H_2O\)). After converting grams of \(O_2\) to moles, we need the mole ratio to convert moles of \(O_2\) to moles of \(H_2O\). Option B has \(\frac{2.0\space mole\space H_2O}{1.0\space mole\space O_2}\), which is the correct mole ratio from the balanced equation. Options A, C, and D do not represent the correct mole ratio or incorrect mass - mole conversion for this step.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. \(\frac{2.0\space mole\space H_2O}{1.0\space mole\space O_2}\)