QUESTION IMAGE
Question
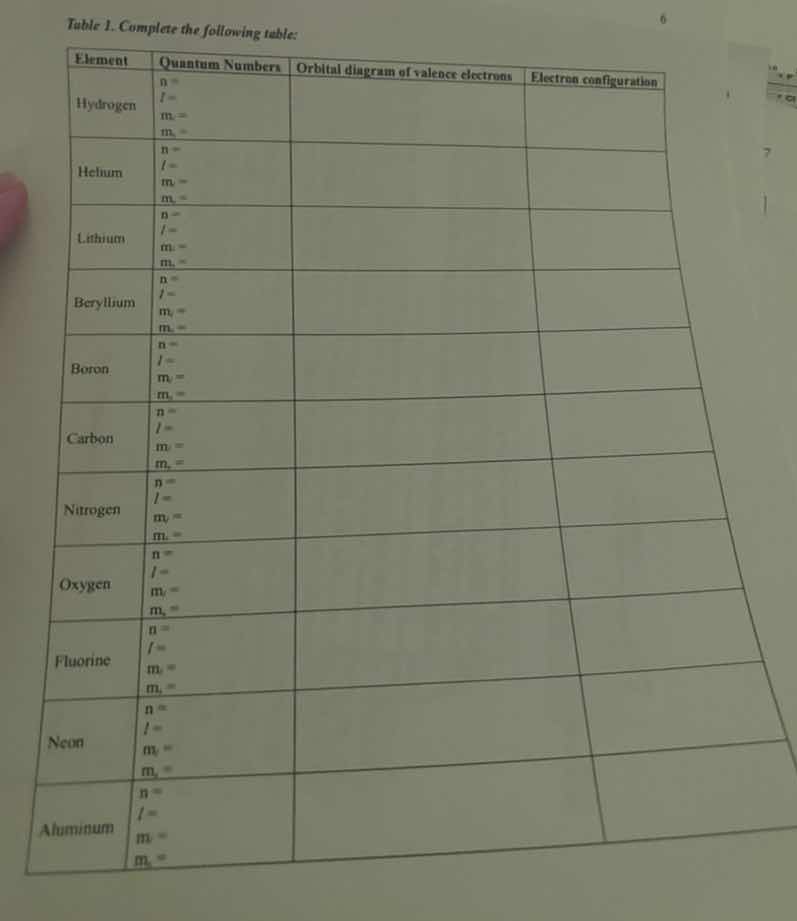
table 1. complete the following table:
| element | quantum numbers | orbital diagram of valence electrons | electron configuration |
|---|---|---|---|
| helium | $n=$<br>$l=$<br>$m_l=$<br>$m_s=$ | ||
| lithium | $n=$<br>$l=$<br>$m_l=$<br>$m_s=$ | ||
| beryllium | $n=$<br>$l=$<br>$m_l=$<br>$m_s=$ | ||
| boron | $n=$<br>$l=$<br>$m_l=$<br>$m_s=$ | ||
| carbon | $n=$<br>$l=$<br>$m_l=$<br>$m_s=$ | ||
| nitrogen | $n=$<br>$l=$<br>$m_l=$<br>$m_s=$ | ||
| oxygen | $n=$<br>$l=$<br>$m_l=$<br>$m_s=$ | ||
| fluorine | $n=$<br>$l=$<br>$m_l=$<br>$m_s=$ | ||
| neon | $n=$<br>$l=$<br>$m_l=$<br>$m_s=$ | ||
| aluminum | $n=$<br>$l=$<br>$m_l=$<br>$m_s=$ |
Step1: Define quantum number rules
- $n$ (principal): shell of valence electrons
- $l$ (azimuthal): $l=0$ for s-orbitals, $l=1$ for p-orbitals
- $m_l$ (magnetic): $m_l=0$ for s; $m_l=-1,0,+1$ for p
- $m_s$ (spin): $+\frac{1}{2}$ or $-\frac{1}{2}$ for electrons
Step2: Fill each element's data
Hydrogen (H, Z=1)
Quantum Numbers:
$n=1$, $l=0$, $m_l=0$, $m_s=+\frac{1}{2}$
Orbital diagram (valence): $\uparrow$ (1s)
Electron configuration: $1s^1$
Helium (He, Z=2)
Quantum Numbers:
$n=1$, $l=0$, $m_l=0$, $m_s=-\frac{1}{2}$ (for 2nd e⁻)
Orbital diagram (valence): $\uparrow\downarrow$ (1s)
Electron configuration: $1s^2$
Lithium (Li, Z=3)
Quantum Numbers (valence e⁻):
$n=2$, $l=0$, $m_l=0$, $m_s=+\frac{1}{2}$
Orbital diagram (valence): $\uparrow$ (2s)
Electron configuration: $1s^22s^1$
Beryllium (Be, Z=4)
Quantum Numbers (valence e⁻):
$n=2$, $l=0$, $m_l=0$, $m_s=-\frac{1}{2}$
Orbital diagram (valence): $\uparrow\downarrow$ (2s)
Electron configuration: $1s^22s^2$
Boron (B, Z=5)
Quantum Numbers (valence e⁻):
$n=2$, $l=1$, $m_l=-1$, $m_s=+\frac{1}{2}$
Orbital diagram (valence): $\uparrow\downarrow$ (2s) $\quad \uparrow$ (2pₓ)
Electron configuration: $1s^22s^22p^1$
Carbon (C, Z=6)
Quantum Numbers (valence e⁻):
$n=2$, $l=1$, $m_l=0$, $m_s=+\frac{1}{2}$
Orbital diagram (valence): $\uparrow\downarrow$ (2s) $\quad \uparrow$ (2pₓ) $\uparrow$ (2pᵧ)
Electron configuration: $1s^22s^22p^2$
Nitrogen (N, Z=7)
Quantum Numbers (valence e⁻):
$n=2$, $l=1$, $m_l=+1$, $m_s=+\frac{1}{2}$
Orbital diagram (valence): $\uparrow\downarrow$ (2s) $\quad \uparrow$ (2pₓ) $\uparrow$ (2pᵧ) $\uparrow$ (2p_z)
Electron configuration: $1s^22s^22p^3$
Oxygen (O, Z=8)
Quantum Numbers (valence e⁻):
$n=2$, $l=1$, $m_l=-1$, $m_s=-\frac{1}{2}$
Orbital diagram (valence): $\uparrow\downarrow$ (2s) $\quad \uparrow\downarrow$ (2pₓ) $\uparrow$ (2pᵧ) $\uparrow$ (2p_z)
Electron configuration: $1s^22s^22p^4$
Fluorine (F, Z=9)
Quantum Numbers (valence e⁻):
$n=2$, $l=1$, $m_l=0$, $m_s=-\frac{1}{2}$
Orbital diagram (valence): $\uparrow\downarrow$ (2s) $\quad \uparrow\downarrow$ (2pₓ) $\uparrow\downarrow$ (2pᵧ) $\uparrow$ (2p_z)
Electron configuration: $1s^22s^22p^5$
Neon (Ne, Z=10)
Quantum Numbers (valence e⁻):
$n=2$, $l=1$, $m_l=+1$, $m_s=-\frac{1}{2}$
Orbital diagram (valence): $\uparrow\downarrow$ (2s) $\quad \uparrow\downarrow$ (2pₓ) $\uparrow\downarrow$ (2pᵧ) $\uparrow\downarrow$ (2p_z)
Electron configuration: $1s^22s^22p^6$
Aluminum (Al, Z=13)
Quantum Numbers (valence e⁻):
$n=3$, $l=1$, $m_l=-1$, $m_s=+\frac{1}{2}$
Orbital diagram (valence): $\uparrow\downarrow$ (3s) $\quad \uparrow$ (3pₓ)
Electron configuration: $1s^22s^22p^63s^23p^1$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Element | Quantum Numbers | Orbital diagram of valence electrons | Electron configuration |
|---|---|---|---|
| Helium | $n=1$, $l=0$, $m_l=0$, $m_s=-\frac{1}{2}$ | $\uparrow\downarrow$ (1s) | $1s^2$ |
| Lithium | $n=2$, $l=0$, $m_l=0$, $m_s=+\frac{1}{2}$ | $\uparrow$ (2s) | $1s^22s^1$ |
| Beryllium | $n=2$, $l=0$, $m_l=0$, $m_s=-\frac{1}{2}$ | $\uparrow\downarrow$ (2s) | $1s^22s^2$ |
| Boron | $n=2$, $l=1$, $m_l=-1$, $m_s=+\frac{1}{2}$ | $\uparrow\downarrow$ (2s) $\quad \uparrow$ (2pₓ) | $1s^22s^22p^1$ |
| Carbon | $n=2$, $l=1$, $m_l=0$, $m_s=+\frac{1}{2}$ | $\uparrow\downarrow$ (2s) $\quad \uparrow$ (2pₓ) $\uparrow$ (2pᵧ) | $1s^22s^22p^2$ |
| Nitrogen | $n=2$, $l=1$, $m_l=+1$, $m_s=+\frac{1}{2}$ | $\uparrow\downarrow$ (2s) $\quad \uparrow$ (2pₓ) $\uparrow$ (2pᵧ) $\uparrow$ (2p_z) | $1s^22s^22p^3$ |
| Oxygen | $n=2$, $l=1$, $m_l=-1$, $m_s=-\frac{1}{2}$ | $\uparrow\downarrow$ (2s) $\quad \uparrow\downarrow$ (2pₓ) $\uparrow$ (2pᵧ) $\uparrow$ (2p_z) | $1s^22s^22p^4$ |
| Fluorine | $n=2$, $l=1$, $m_l=0$, $m_s=-\frac{1}{2}$ | $\uparrow\downarrow$ (2s) $\quad \uparrow\downarrow$ (2pₓ) $\uparrow\downarrow$ (2pᵧ) $\uparrow$ (2p_z) | $1s^22s^22p^5$ |
| Neon | $n=2$, $l=1$, $m_l=+1$, $m_s=-\frac{1}{2}$ | $\uparrow\downarrow$ (2s) $\quad \uparrow\downarrow$ (2pₓ) $\uparrow\downarrow$ (2pᵧ) $\uparrow\downarrow$ (2p_z) | $1s^22s^22p^6$ |
| Aluminum | $n=3$, $l=1$, $m_l=-1$, $m_s=+\frac{1}{2}$ | $\uparrow\downarrow$ (3s) $\quad \uparrow$ (3pₓ) | $1s^22s^22p^63s^23p^1$ |