QUESTION IMAGE
Question
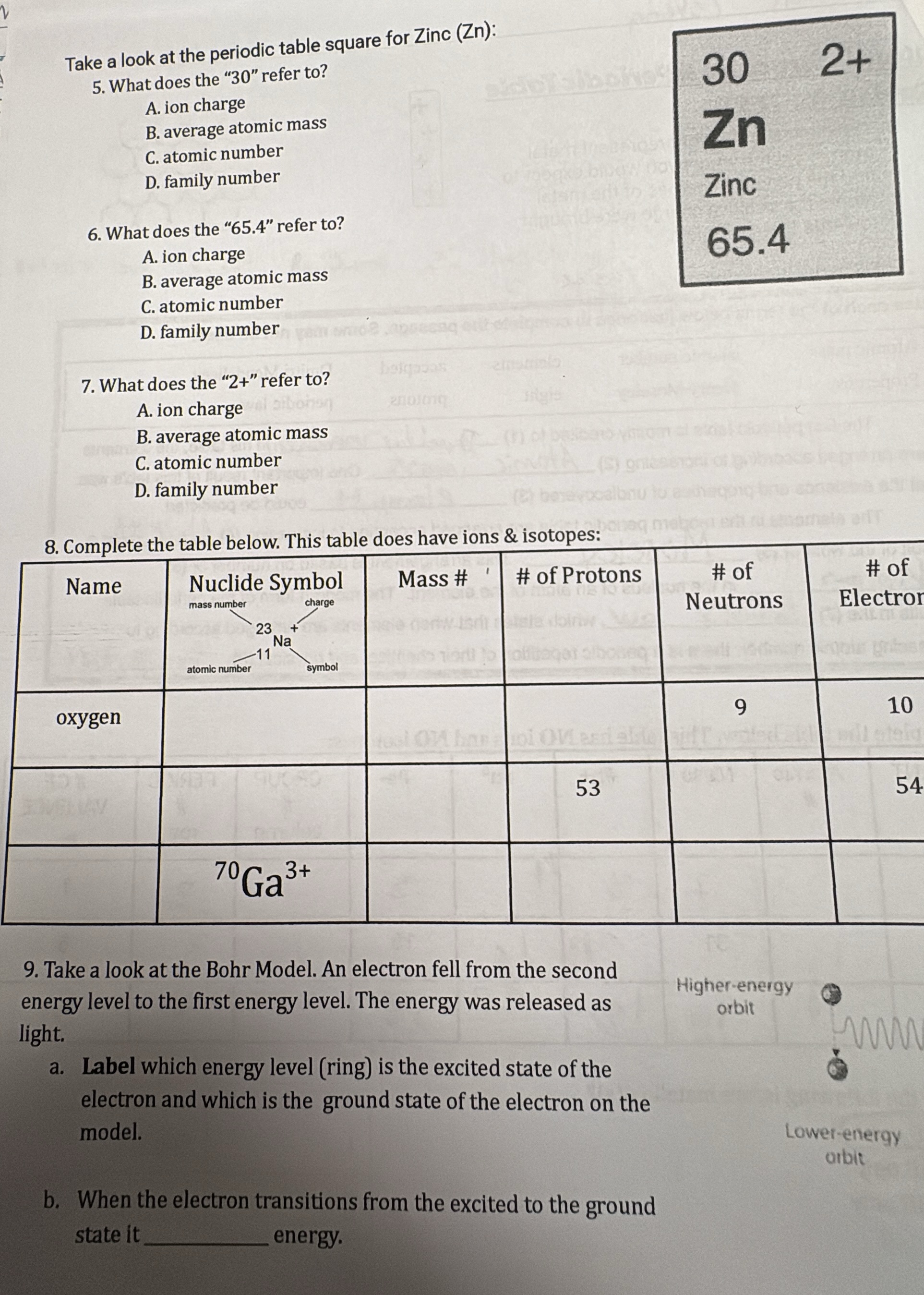
take a look at the periodic table square for zinc (zn):
- what does the “30” refer to?
a. ion charge
b. average atomic mass
c. atomic number
d. family number
- what does the “65.4” refer to?
a. ion charge
b. average atomic mass
c. atomic number
d. family number
- what does the “2+” refer to?
a. ion charge
b. average atomic mass
c. atomic number
d. family number
- complete the table below. this table does have ions & isotopes:
| name | nuclide symbol (with mass number, atomic number, charge, symbol labeled as in ²³₁₁na⁺) | mass # | # of protons | # of neutrons | # of electrons |
|---|---|---|---|---|---|
| 53 | 54 | ||||
| ⁷⁰ga³⁺ |
- take a look at the bohr model. an electron fell from the second energy level to the first energy level. the energy was released as light.
a. label which energy level (ring) is the excited state of the electron and which is the ground state of the electron on the model (with an image showing higher - energy orbit and lower - energy orbit with an electron transition).
b. when the electron transitions from the excited to the ground state it ______ energy.
Question 5
In a periodic table square, the atomic number is the number of protons in an atom's nucleus, and it's typically the whole number at the top. For Zinc, "30" is the atomic number (number of protons), while ion charge is "2+", average atomic mass is "65.4", and family number isn't represented this way. So "30" refers to atomic number.
In the periodic table square, the number at the bottom (usually a decimal) represents the average atomic mass of the element, which is the weighted average of the masses of its isotopes. "65.4" fits this description; ion charge is "2+", atomic number is "30", and family number isn't this. So "65.4" is average atomic mass.
The "2+" in the Zinc square indicates the charge of the Zinc ion (how many electrons it has gained or lost). Ion charge is represented by a symbol with a sign and number, while average atomic mass is "65.4", atomic number is "30", and family number isn't this. So "2+" is ion charge.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. atomic number