QUESTION IMAGE
Question
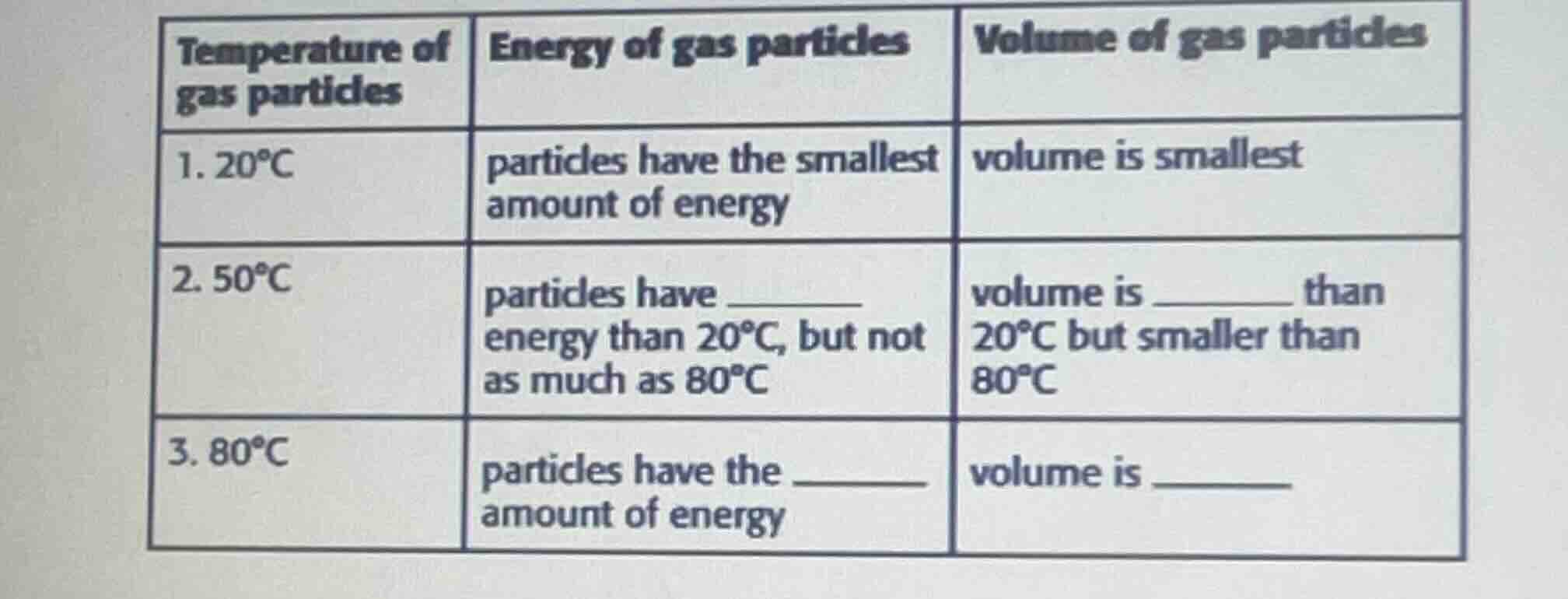
temperature of gas particles | energy of gas particles | volume of gas particles
- 20°c | particles have the smallest amount of energy | volume is smallest
- 50°c | particles have ____ energy than 20°c, but not as much as 80°c | volume is ____ than 20°c but smaller than 80°c
- 80°c | particles have the ____ amount of energy | volume is ____
Brief Explanations
For gas particles, temperature is directly related to particle kinetic energy and volume (per Charles's Law, assuming constant pressure). Higher temperature means higher particle energy and larger volume.
- At 50°C, which is between 20°C and 80°C, particle energy is greater than at 20°C but less than at 80°C, and volume is larger than at 20°C but smaller than at 80°C.
- At 80°C (the highest temperature listed), particles have the greatest amount of energy, and the volume is the largest.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- 20°C: (filled as given)
- 50°C:
- Energy of gas particles: particles have more energy than 20°C, but not as much as 80°C
- Volume of gas particles: volume is larger than 20°C but smaller than 80°C
- 80°C:
- Energy of gas particles: particles have the largest/greatest amount of energy
- Volume of gas particles: volume is the largest