QUESTION IMAGE
Question
ts elementary : building elements
chapter pages
tool: interactive periodic t
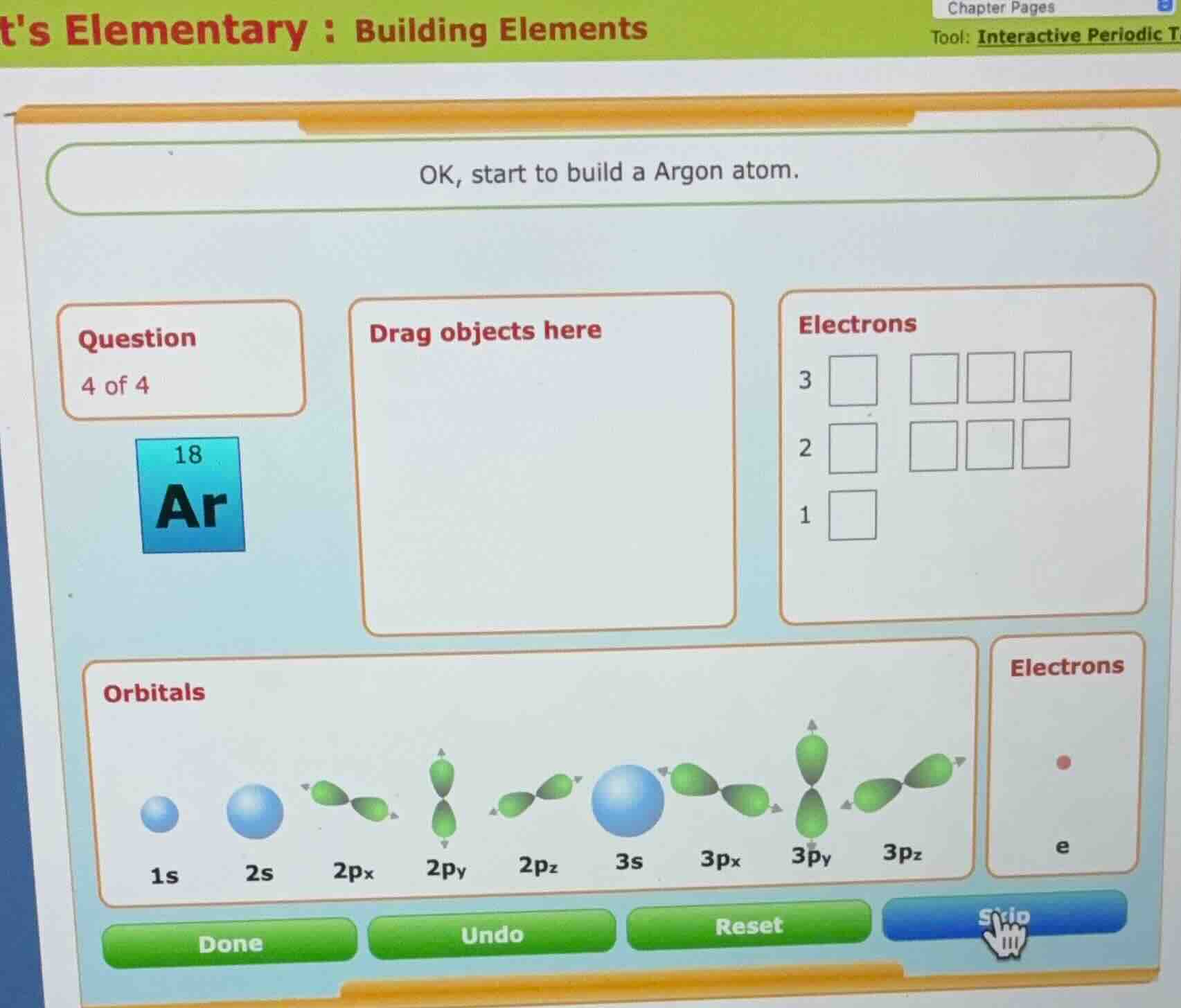
ok, start to build a argon atom.
question
4 of 4
18
ar
drag objects here
electrons
3
2
1
orbitals
1s 2s 2pₓ 2pᵧ 2p_z 3s 3pₓ 3pᵧ 3p_z
electrons
e
done undo reset skip
Step1: Identify atomic number of Ar
Argon (Ar) has an atomic number of 18, so it has 18 electrons.
Step2: Fill orbitals by Aufbau principle
Fill lower energy orbitals first:
- 1s orbital holds 2 electrons: $1s^2$
- 2s orbital holds 2 electrons: $2s^2$
- 2p orbitals ($2p_x, 2p_y, 2p_z$) each hold 2 electrons: $2p^6$
- 3s orbital holds 2 electrons: $3s^2$
- 3p orbitals ($3p_x, 3p_y, 3p_z$) each hold 2 electrons: $3p^6$
Verify total electrons: $2+2+6+2+6=18$
Step3: Map to the interface
Drag the following orbitals to the "Drag objects here" box, then add 2 electrons to each:
- 1s, 2s, 2p$_x$, 2p$_y$, 2p$_z$, 3s, 3p$_x$, 3p$_y$, 3p$_z$
- Assign electrons:
- Row 1 (1s): 2 electrons
- Row 2 (2s, 2p$_x$, 2p$_y$, 2p$_z$): 2 electrons each (total 8)
- Row 3 (3s, 3p$_x$, 3p$_y$, 3p$_z$): 2 electrons each (total 8)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Drag all orbitals (1s, 2s, 2p$_x$, 2p$_y$, 2p$_z$, 3s, 3p$_x$, 3p$_y$, 3p$_z$) to the "Drag objects here" area.
- Assign electrons as follows:
- Row 1 (1s): Fill the box with 2 electrons
- Row 2 (2s, 2p$_x$, 2p$_y$, 2p$_z$): Fill each box with 2 electrons
- Row 3 (3s, 3p$_x$, 3p$_y$, 3p$_z$): Fill each box with 2 electrons
Total electrons: 18, matching Argon's electron count.