QUESTION IMAGE
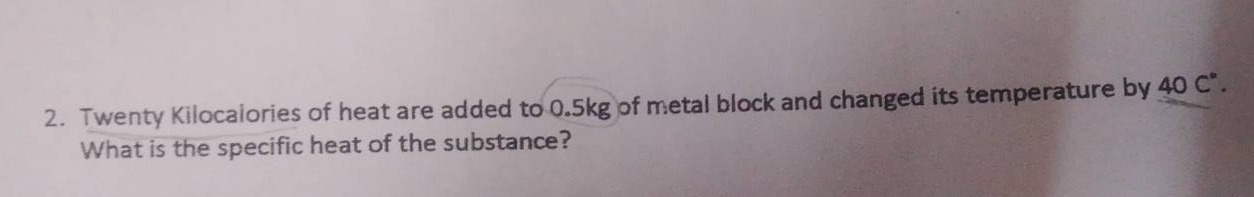
Question
- twenty kilocalories of heat are added to 0.5kg of metal block and changed its temperature by 40 °c. what is the specific heat of the substance?
Step1: Recall the heat formula
The formula for heat energy is \( Q = mc\Delta T \), where \( Q \) is the heat energy, \( m \) is the mass, \( c \) is the specific heat, and \( \Delta T \) is the change in temperature. We need to solve for \( c \), so rearrange the formula to \( c=\frac{Q}{m\Delta T} \).
Step2: Convert units (if necessary)
First, convert \( Q = 20 \) Kilocalories to calories (since 1 Kilocalorie = 1000 calories), so \( Q = 20\times1000 = 20000 \) calories. The mass \( m = 0.5 \) kg = \( 0.5\times1000 = 500 \) g (we can use grams here as the unit for mass in the calorie - gram - Celsius system, where the specific heat unit will be cal/(g·°C)). The change in temperature \( \Delta T = 40^{\circ}\text{C} \).
Step3: Substitute values into the formula
Substitute \( Q = 20000 \) cal, \( m = 500 \) g, and \( \Delta T = 40^{\circ}\text{C} \) into the formula \( c=\frac{Q}{m\Delta T} \).
\( c=\frac{20000}{500\times40} \)
First, calculate the denominator: \( 500\times40 = 20000 \)
Then, \( c=\frac{20000}{20000}=1 \) cal/(g·°C)
We can also use SI units. Convert \( Q = 20 \) Kilocalories to joules. We know that 1 calorie = 4.184 joules, so 1 Kilocalorie = 4184 joules. Then \( Q = 20\times4184=83680 \) J. The mass \( m = 0.5 \) kg, \( \Delta T = 40 \) K (since a change in Celsius is the same as a change in Kelvin). Using the formula \( Q = mc\Delta T \) (in SI units, \( c \) has units of J/(kg·K)). Rearranging for \( c \), \( c=\frac{Q}{m\Delta T} \)
Substitute \( Q = 83680 \) J, \( m = 0.5 \) kg, \( \Delta T = 40 \) K
\( c=\frac{83680}{0.5\times40}=\frac{83680}{20} = 4184 \) J/(kg·K) (which is consistent with the specific heat of water, but in this case, the metal has a specific heat of 1 cal/(g·°C) or 4184 J/(kg·K))
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The specific heat of the substance is \( 1 \) cal/(g·°C) (or \( 4184 \) J/(kg·K))