QUESTION IMAGE
Question
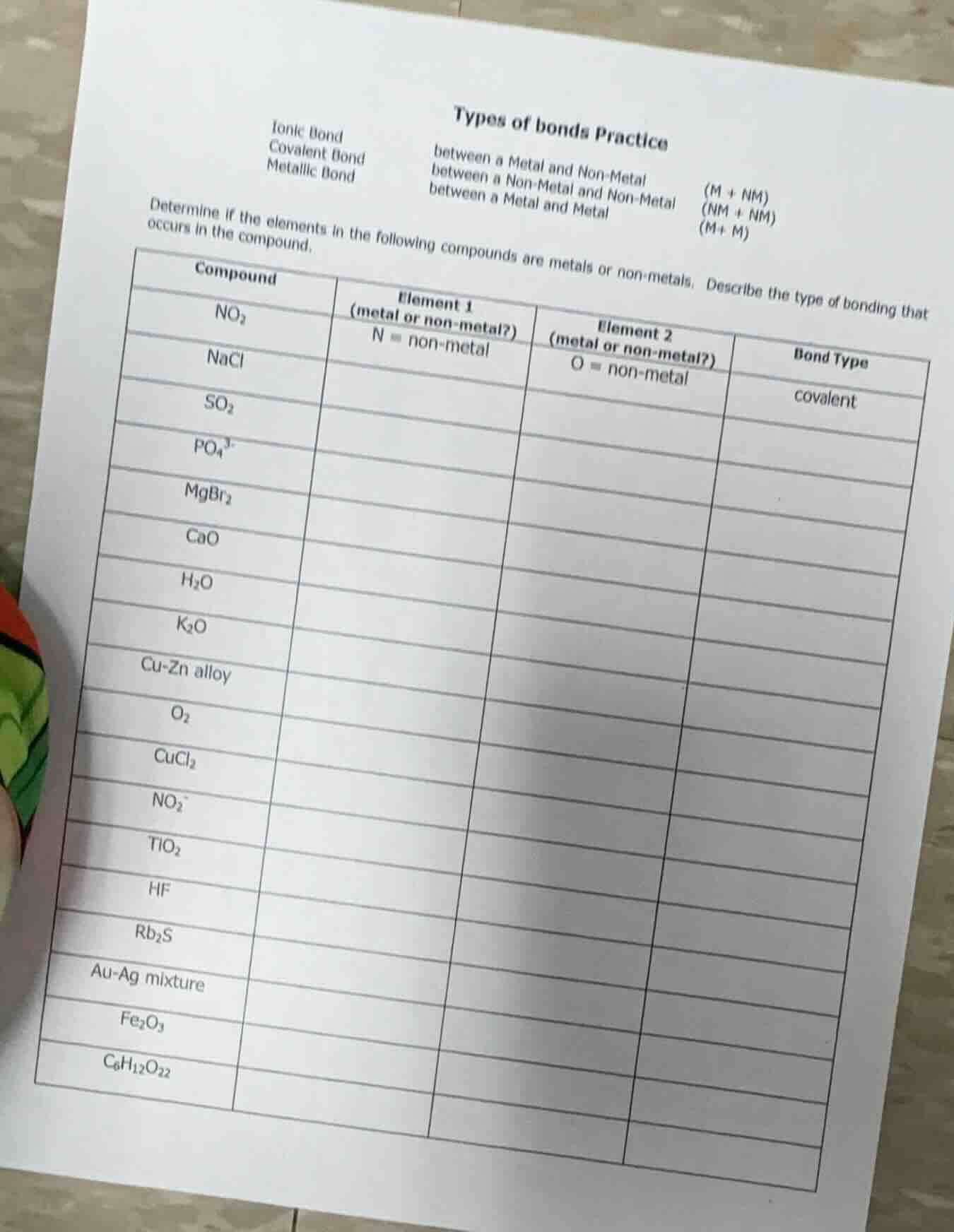
types of bonds practice
ionic bond between a metal and non-metal (m + nm)
covalent bond between a non-metal and non-metal (nm + nm)
metallic bond between a metal and metal (m+ m)
determine if the elements in the following compounds are metals or non-metals. describe the type of bonding that occurs in the compound.
| compound | element 1 (metal or non-metal?) | element 2 (metal or non-metal?) | bond type | ---- | ---- | ---- | ---- | $\text{no}_2$ | n = non-metal | o = non-metal | covalent | $\text{nacl}$ | $\text{so}_2$ | $\text{po}_4^{3-}$ | $\text{mgbr}_2$ | $\text{cao}$ | $\text{h}_2\text{o}$ | $\text{k}_2\text{o}$ | cu-zn alloy | $\text{o}_2$ | $\text{cucl}_2$ | $\text{no}_2^-$ | $\text{tio}_2$ | $\text{hf}$ | $\text{rb}_2\text{s}$ | au-ag mixture | $\text{fe}_2\text{o}_3$ | $\text{c}_6\text{h}_{12}\text{o}_{22}$ |
Step1: Classify elements, assign bond type
For each compound, identify if elements are metal (M) or non-metal (NM), then match to bond rules: ionic (M+NM), covalent (NM+NM), metallic (M+M).
Step2: Fill table systematically
Go through each compound one by one, apply classification rules.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Compound | Element 1 (metal or non-metal?) | Element 2 (metal or non-metal?) | Bond Type |
|---|---|---|---|
| $\text{NaCl}$ | Na = metal | Cl = non-metal | ionic |
| $\text{SO}_2$ | S = non-metal | O = non-metal | covalent |
| $\text{PO}_4^{3-}$ | P = non-metal | O = non-metal | covalent |
| $\text{MgBr}_2$ | Mg = metal | Br = non-metal | ionic |
| $\text{CaO}$ | Ca = metal | O = non-metal | ionic |
| $\text{H}_2\text{O}$ | H = non-metal | O = non-metal | covalent |
| $\text{K}_2\text{O}$ | K = metal | O = non-metal | ionic |
| Cu-Zn alloy | Cu = metal | Zn = metal | metallic |
| $\text{O}_2$ | O = non-metal | O = non-metal | covalent |
| $\text{CuCl}_2$ | Cu = metal | Cl = non-metal | ionic |
| $\text{NO}_2^-$ | N = non-metal | O = non-metal | covalent |
| $\text{TiO}_2$ | Ti = metal | O = non-metal | ionic |
| $\text{HF}$ | H = non-metal | F = non-metal | covalent |
| $\text{Rb}_2\text{S}$ | Rb = metal | S = non-metal | ionic |
| Au-Ag mixture | Au = metal | Ag = metal | metallic |
| $\text{Fe}_2\text{O}_3$ | Fe = metal | O = non-metal | ionic |
| $\text{C}_{6}\text{H}_{12}\text{O}_{22}$ | C = non-metal, H = non-metal | O = non-metal | covalent |