QUESTION IMAGE
Question
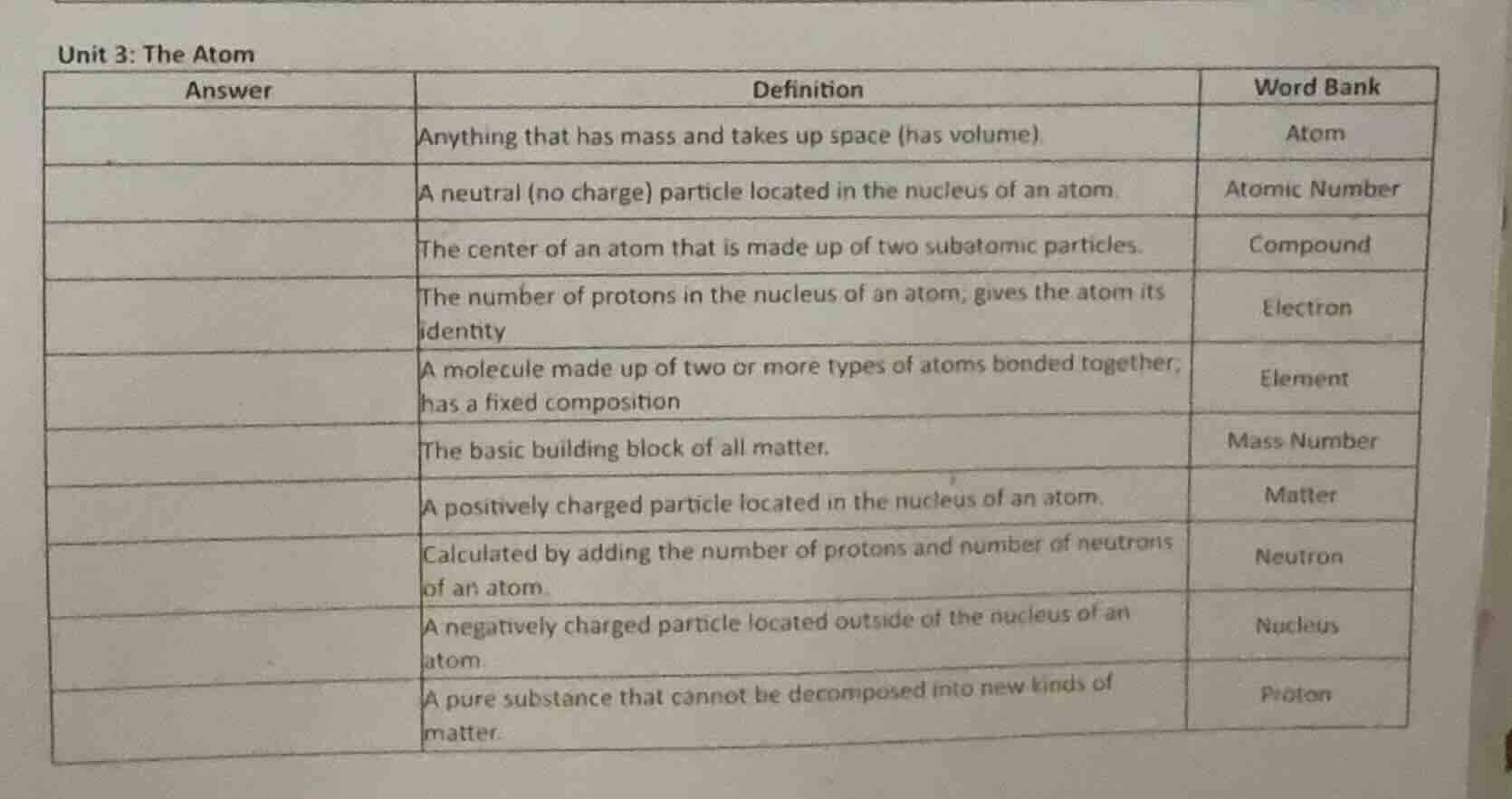
unit 3: the atom
| answer | definition | word bank |
|---|---|---|
| a neutral (no charge) particle located in the nucleus of an atom | atomic number | |
| the center of an atom that is made up of two subatomic particles. | compound | |
| the number of protons in the nucleus of an atom; gives the atom its identity | electron | |
| a molecule made up of two or more types of atoms bonded together; has a fixed composition | element | |
| the basic building block of all matter. | mass number | |
| a positively charged particle located in the nucleus of an atom. | matter | |
| calculated by adding the number of protons and number of neutrons of an atom. | neutron | |
| a negatively charged particle located outside of the nucleus of an atom. | nucleus | |
| a pure substance that cannot be decomposed into new kinds of matter. | proton |
Brief Explanations
Match each definition to the correct term from the word bank by identifying the key traits in each definition (e.g., charge, location, composition, or core function of the particle/substance).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Answer | Definition |
|---|---|
| Neutron | A neutral (no charge) particle located in the nucleus of an atom. |
| Nucleus | The center of an atom that is made up of two subatomic particles. |
| Atomic Number | The number of protons in the nucleus of an atom; gives the atom its identity |
| Compound | A molecule made up of two or more types of atoms bonded together; has a fixed composition |
| Atom | The basic building block of all matter. |
| Proton | A positively charged particle located in the nucleus of an atom. |
| Mass Number | Calculated by adding the number of protons and number of neutrons of an atom. |
| Electron | A negatively charged particle located outside of the nucleus of an atom. |
| Element | A pure substance that cannot be decomposed into new kinds of matter. |