QUESTION IMAGE
Question
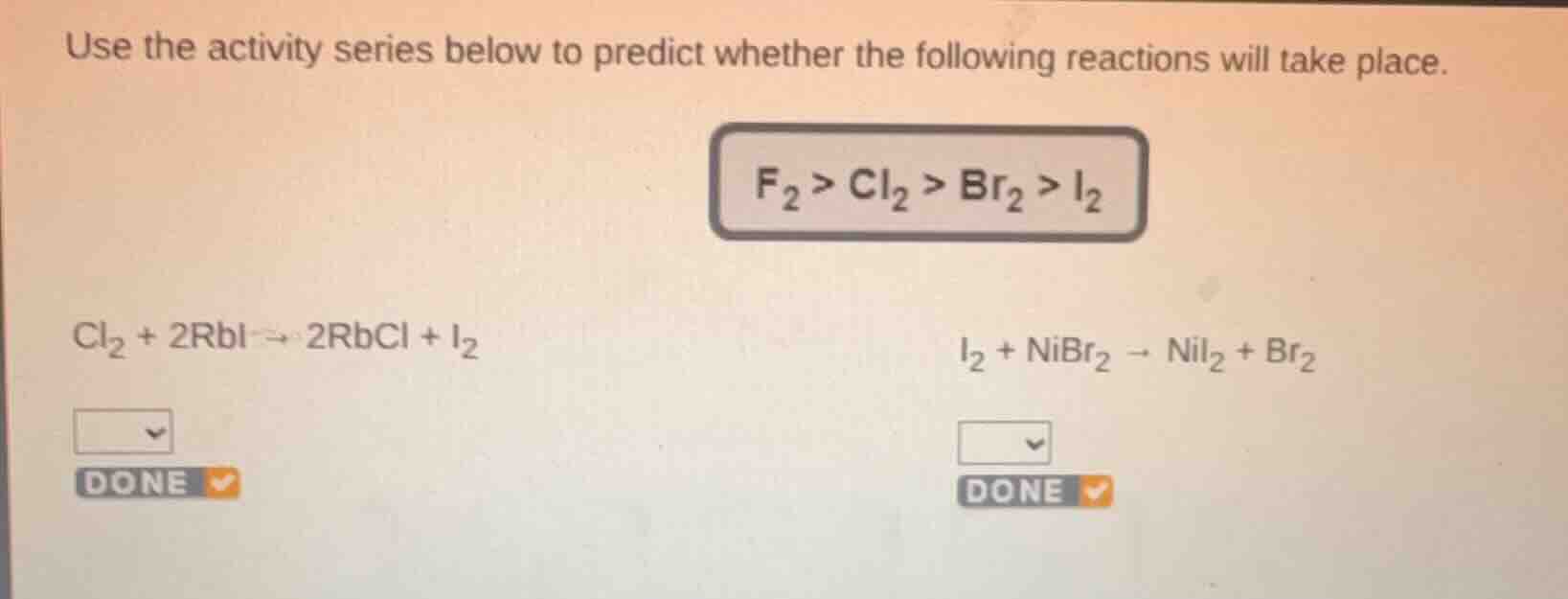
use the activity series below to predict whether the following reactions will take place.
f₂ > cl₂ > br₂ > i₂
cl₂ + 2rbi → 2rbcl + i₂
i₂ + nibr₂ → nii₂ + br₂
for First Reaction ($\mathbf{Cl_2 + 2RbI
ightarrow 2RbCl + I_2}$):
Step1: Check Activity Series Position
The activity series is $F_2 > Cl_2 > Br_2 > I_2$. Here, $Cl_2$ is more reactive than $I_2$ (since $Cl_2$ comes before $I_2$ in the series).
Step2: Determine Reaction Feasibility
In a single - replacement reaction (displacement reaction) involving halogens, a more reactive halogen can displace a less reactive halogen from its compound. Since $Cl_2$ is more reactive than $I_2$, $Cl_2$ can displace $I^-$ from $RbI$ to form $RbCl$ and $I_2$. So, this reaction will take place.
for Second Reaction ($\mathbf{I_2 + NiBr_2
ightarrow NiI_2 + Br_2}$):
Step1: Check Activity Series Position
From the activity series $F_2 > Cl_2 > Br_2 > I_2$, we see that $I_2$ is less reactive than $Br_2$ (because $Br_2$ comes before $I_2$ in the series).
Step2: Determine Reaction Feasibility
For a halogen displacement reaction, a less reactive halogen cannot displace a more reactive halogen from its compound. Since $I_2$ is less reactive than $Br_2$, $I_2$ cannot displace $Br^-$ from $NiBr_2$ to form $NiI_2$ and $Br_2$. So, this reaction will not take place.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s:
- For the reaction $\boldsymbol{Cl_2 + 2RbI
ightarrow 2RbCl + I_2}$: The reaction will take place.
- For the reaction $\boldsymbol{I_2 + NiBr_2
ightarrow NiI_2 + Br_2}$: The reaction will not take place.