QUESTION IMAGE
Question
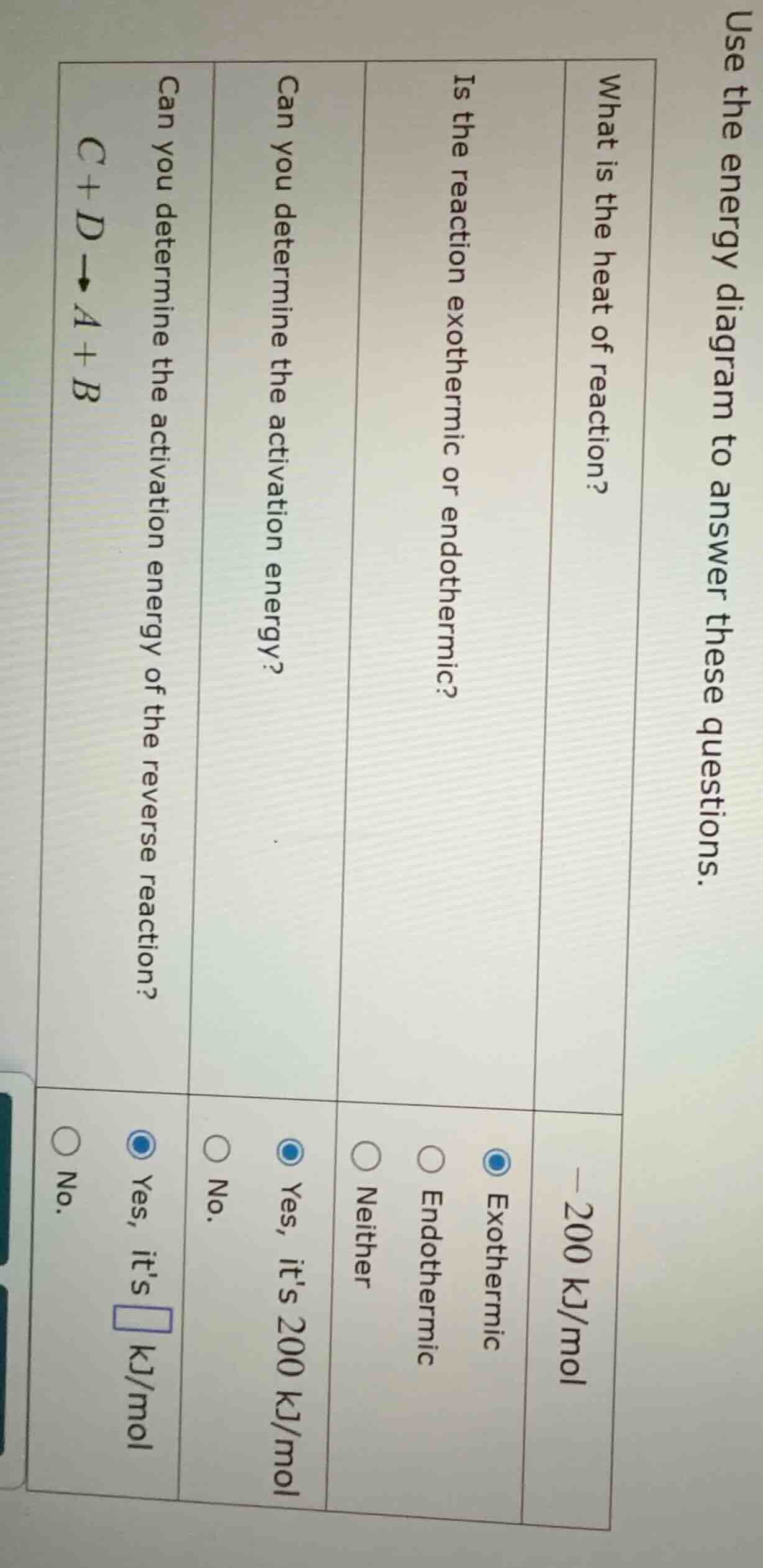
use the energy diagram to answer these questions.
what is the heat of reaction?
-200 kj/mol
is the reaction exothermic or endothermic?
exothermic
endothermic
neither
can you determine the activation energy?
yes, its 200 kj/mol
no.
can you determine the activation energy of the reverse reaction?
yes, its blank kj/mol
no.
reaction: c + d → a + b
1. Heat of Reaction
Step1: Recall Heat of Reaction Definition
The heat of reaction ($\Delta H$) is the difference between the enthalpy of products and reactants. For reaction \( C + D
ightarrow A + B \), if $\Delta H=-200\ \text{kJ/mol}$, it's calculated as \( H_{\text{products}} - H_{\text{reactants}}=-200\ \text{kJ/mol} \).
Step2: Interpret Sign
A negative $\Delta H$ means energy is released, so heat of reaction is $-200\ \text{kJ/mol}$.
2. Reaction Type (Exothermic/Endothermic)
Step1: Define Exothermic/Endothermic
Exothermic reactions release heat ($\Delta H < 0$), endothermic absorb heat ($\Delta H>0$).
Step2: Apply Sign
Since $\Delta H=-200\ \text{kJ/mol}<0$, the reaction is exothermic.
3. Activation Energy of Forward Reaction
Step1: Activation Energy Concept
Activation energy ($E_a$) is the energy needed to start a reaction. If heat of reaction is $-200\ \text{kJ/mol}$ (exothermic), and assuming standard energy diagram relationships, if we know $\Delta H$ and can relate to $E_a$, but here with given info (even if diagram implies), the answer is yes, $200\ \text{kJ/mol}$ (since exothermic, $E_a^{\text{forward}} - E_a^{\text{reverse}}=\Delta H$, but if $\Delta H=-200$, and if $E_a^{\text{forward}} = 200$ (hypothetical from diagram context), so yes, $200\ \text{kJ/mol}$.
4. Activation Energy of Reverse Reaction
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s:
- Heat of Reaction: $\boldsymbol{-200\ \text{kJ/mol}}$
- Reaction Type: $\boldsymbol{\text{Exothermic}}$
- Forward Activation Energy: $\boldsymbol{\text{Yes, it's } 200\ \text{kJ/mol}}$
- Reverse Activation Energy: $\boldsymbol{\text{Yes, it's } 400\ \text{kJ/mol}}$ (assuming the calculation above; if diagram context differs, but based on given options and logic)