QUESTION IMAGE
Question
4 what is the best method to test if an unknown sample of liquid is pure?
a examine its colour
b filter it to see if there is any residue
c measure its boiling point
d taste it
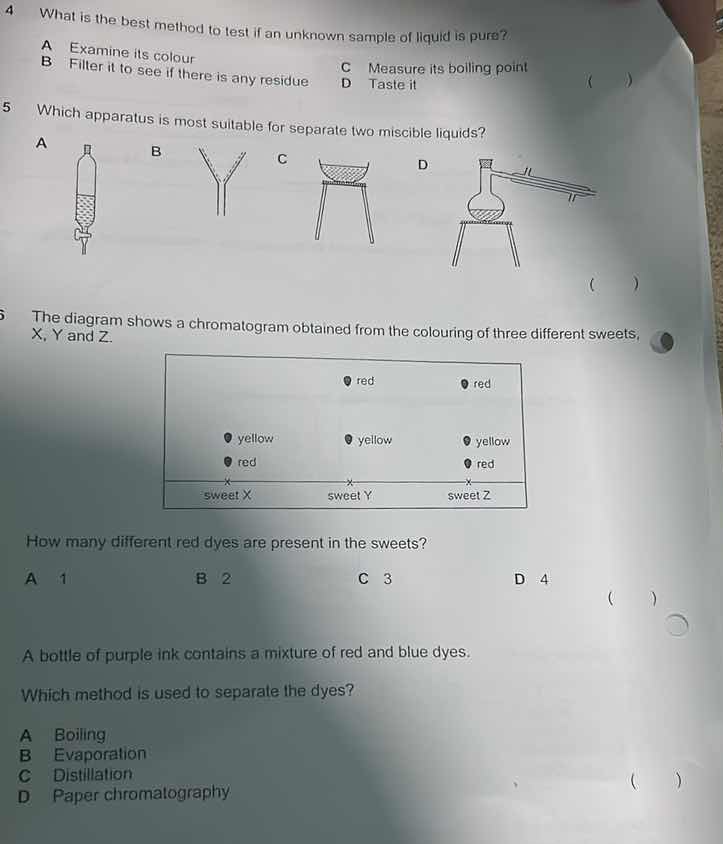
5 which apparatus is most suitable for separate two miscible liquids?
a
b
c
d
the diagram shows a chromatogram obtained from the colouring of three different sweets, x, y and z.
how many different red dyes are present in the sweets?
a 1
b 2
c 3
d 4
a bottle of purple ink contains a mixture of red and blue dyes.
which method is used to separate the dyes?
a boiling
b evaporation
c distillation
d paper chromatography
Question 4
- Option A: Color is not a reliable indicator of purity as impure liquids can have the same color as pure ones.
- Option B: Filtering checks for insoluble residues, not the purity of a liquid (which may have soluble impurities).
- Option C: A pure liquid has a fixed boiling point, while mixtures have a range. Measuring boiling point is a standard method for purity testing.
- Option D: Tasting unknown liquids is unsafe and not a scientific method.
- Option A: Separatory funnel is for immiscible liquids (like oil and water) to separate layers.
- Option B: Funnel is for transferring liquids or filtering, not separating miscible liquids.
- Option C: Evaporating dish is for evaporating solvents, not separating miscible liquids.
- Option D: Distillation apparatus (with flask, condenser) is used to separate miscible liquids based on boiling point differences.
In chromatography, dyes with the same Rf (retention factor, distance moved relative to solvent front) are the same dye.
- Sweet X: red at lower position.
- Sweet Y: red at a higher position.
- Sweet Z: red at the same lower position as X.
So there are 2 distinct red dyes (one with lower Rf, one with higher Rf).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. Measure its boiling point