QUESTION IMAGE
Question
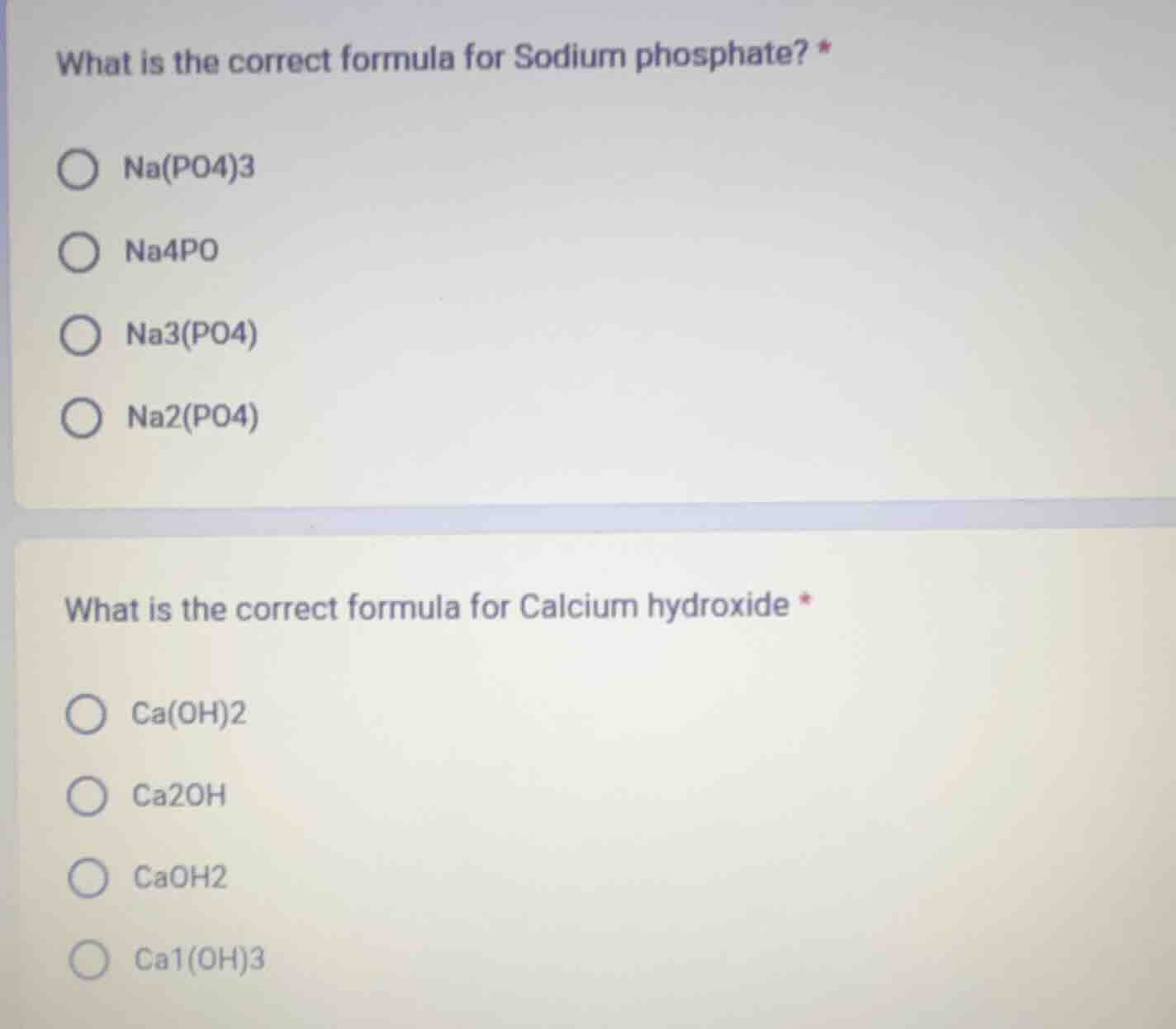
what is the correct formula for sodium phosphate? *
na(po4)3
na4po
na3(po4)
na2(po4)
what is the correct formula for calcium hydroxide *
ca(oh)2
ca2oh
caoh2
ca1(oh)3
First Sub - Question: What is the correct formula for Sodium phosphate?
To determine the formula of sodium phosphate, we use the rules of ionic bonding. Sodium (Na) has a +1 charge, and the phosphate ion ($\text{PO}_4^{3 - }$) has a - 3 charge. To balance the charges, we need 3 sodium ions (each with +1) to balance the - 3 charge of one phosphate ion. So the formula should be $\text{Na}_3\text{PO}_4$ (or $\text{Na}_3(\text{PO}_4)$ as in the option).
Calcium (Ca) has a +2 charge, and the hydroxide ion ($\text{OH}^ - $) has a - 1 charge. To balance the charges, we need 2 hydroxide ions (each with - 1) to balance the +2 charge of one calcium ion. So the formula is $\text{Ca(OH)}_2$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. $\text{Na}_3(\text{PO}_4)$ (assuming the options are labeled as A. $\text{Na(PO}_4\text{)}_3$, B. $\text{Na}_4\text{PO}$, C. $\text{Na}_3(\text{PO}_4)$, D. $\text{Na}_2(\text{PO}_4)$)