QUESTION IMAGE
Question
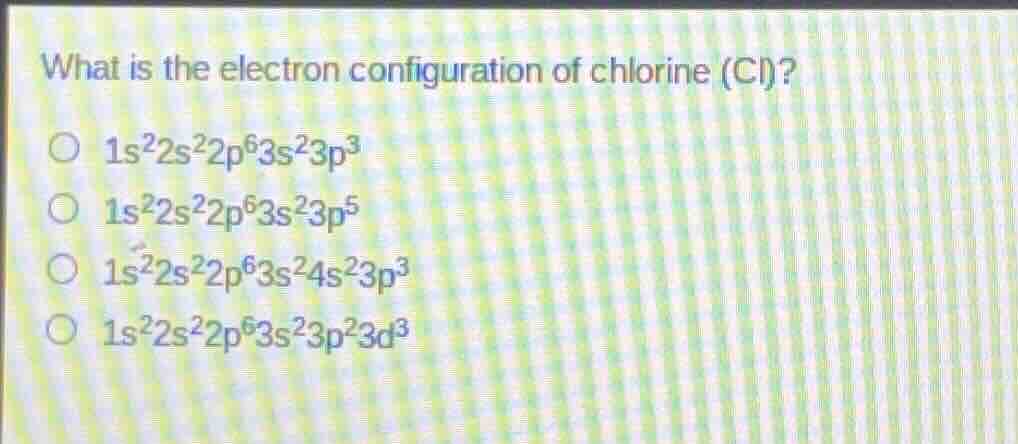
what is the electron configuration of chlorine (cl)?
○ $1s^22s^22p^63s^23p^3$
○ $1s^22s^22p^63s^23p^5$
○ $1s^22s^22p^63s^24s^23p^3$
○ $1s^22s^22p^63s^23p^23d^3$
Brief Explanations
Chlorine (Cl) has an atomic number of 17, meaning it has 17 electrons. The electron configuration follows the Aufbau principle, filling orbitals in order of increasing energy: 1s, 2s, 2p, 3s, 3p, etc.
- The first two electrons go to 1s: \(1s^2\).
- Next two to 2s: \(2s^2\).
- Then six to 2p (since p orbitals can hold 6 electrons): \(2p^6\).
- Next two to 3s: \(3s^2\).
- Remaining \(17 - 2 - 2 - 6 - 2 = 5\) electrons go to 3p: \(3p^5\).
So the electron configuration is \(1s^22s^22p^63s^23p^5\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. \(1s^22s^22p^63s^23p^5\)