QUESTION IMAGE
Question
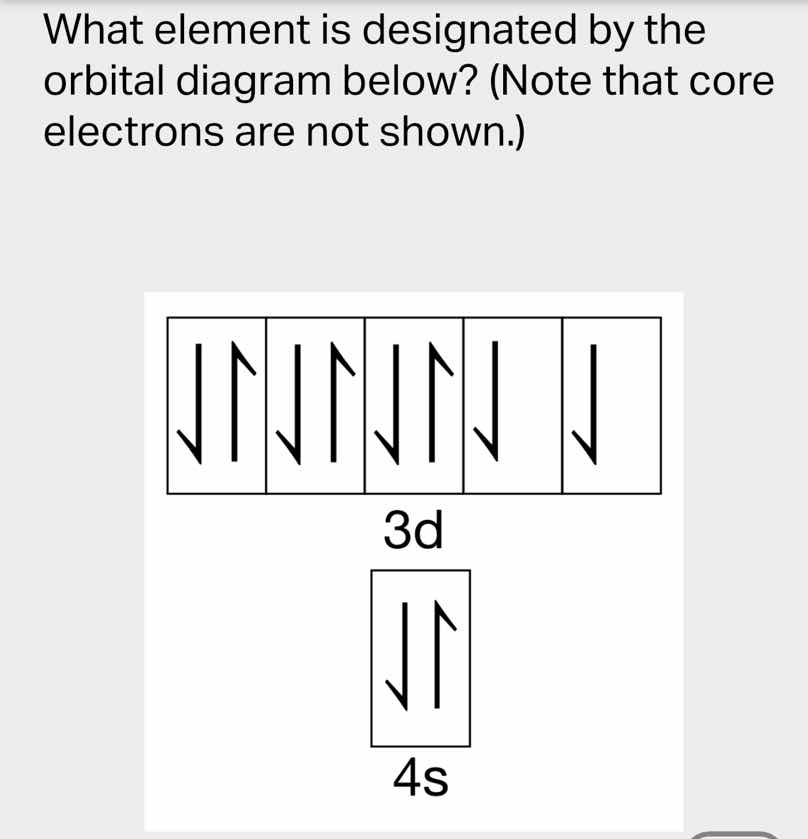
what element is designated by the orbital diagram below? (note that core electrons are not shown.) 3d 4s
Step1: Count valence electrons
Count electrons in 3d and 4s: $3d^8 + 4s^2 = 10$ valence electrons
Step2: Find total electron count
Assume neutral atom: core electrons + valence electrons = [Kr core] + 10 = 36 + 10 = 46
Step3: Match to element
Atomic number 46 corresponds to Palladium? No, correct: 4s²3d⁸ is Nickel? No, wait: 36 (Kr) + 10 = 46 is Pd, but Pd has 4d¹⁰. Wait, no: 4s²3d⁸ is atomic number 28 (Ar core 18 + 10 = 28). Correct: Ar (18) + 2+8=28.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Nickel (Ni)