QUESTION IMAGE
Question
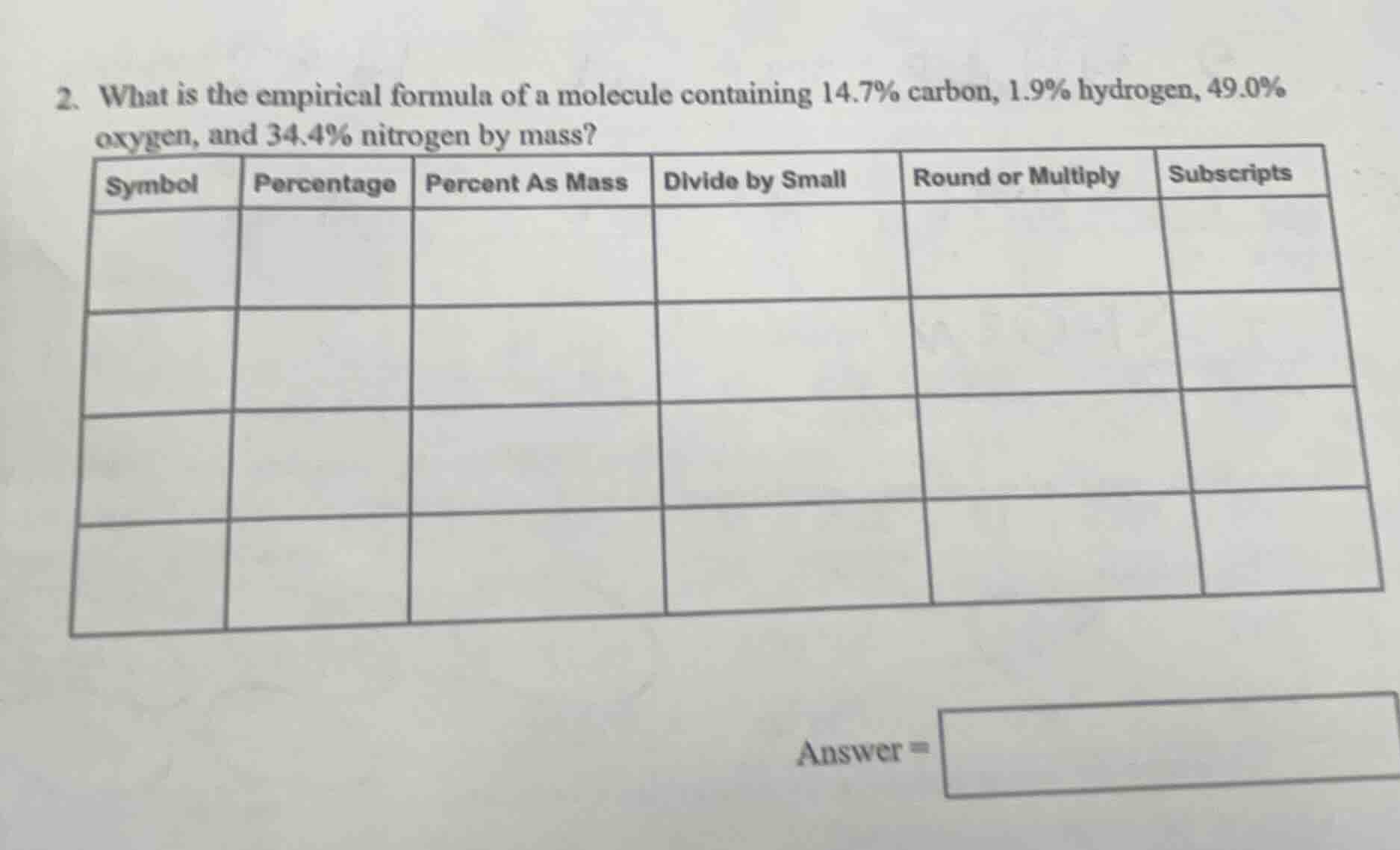
- what is the empirical formula of a molecule containing 14.7% carbon, 1.9% hydrogen, 49.0% oxygen, and 34.4% nitrogen by mass?
symbol | percentage | percent as mass | divide by small | round or multiply | subscripts
answer =
Step1: Assume 100g sample, set masses
Let total mass = 100g. So:
$m_C = 14.7\ \text{g}$, $m_H = 1.9\ \text{g}$, $m_O = 49.0\ \text{g}$, $m_N = 34.4\ \text{g}$
Step2: Calculate moles of each element
Moles $n = \frac{\text{mass}}{\text{molar mass}}$, molar masses: $M_C=12.01\ \text{g/mol}$, $M_H=1.008\ \text{g/mol}$, $M_O=16.00\ \text{g/mol}$, $M_N=14.01\ \text{g/mol}$
$n_C = \frac{14.7}{12.01} \approx 1.224\ \text{mol}$
$n_H = \frac{1.9}{1.008} \approx 1.885\ \text{mol}$
$n_O = \frac{49.0}{16.00} = 3.0625\ \text{mol}$
$n_N = \frac{34.4}{14.01} \approx 2.455\ \text{mol}$
Step3: Divide by smallest mole value
Smallest $n = 1.224\ \text{mol}$
$C: \frac{1.224}{1.224} = 1$
$H: \frac{1.885}{1.224} \approx 1.54$
$O: \frac{3.0625}{1.224} \approx 2.50$
$N: \frac{2.455}{1.224} \approx 2.006$
Step4: Multiply to get whole numbers
Multiply all by 2 to eliminate decimals:
$C: 1 \times 2 = 2$
$H: 1.54 \times 2 \approx 3$
$O: 2.50 \times 2 = 5$
$N: 2.006 \times 2 \approx 4$
Completed Table:
| Symbol | Percentage | Percent As Mass | Divide by Small | Round or Multiply | Subscripts |
|---|---|---|---|---|---|
| H | 1.9% | 1.9 g | ≈1.54 | ×2 | 3 |
| O | 49.0% | 49.0 g | ≈2.50 | ×2 | 5 |
| N | 34.4% | 34.4 g | ≈2.006 | ×2 | 4 |
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\text{C}_2\text{H}_3\text{O}_5\text{N}_4$