QUESTION IMAGE
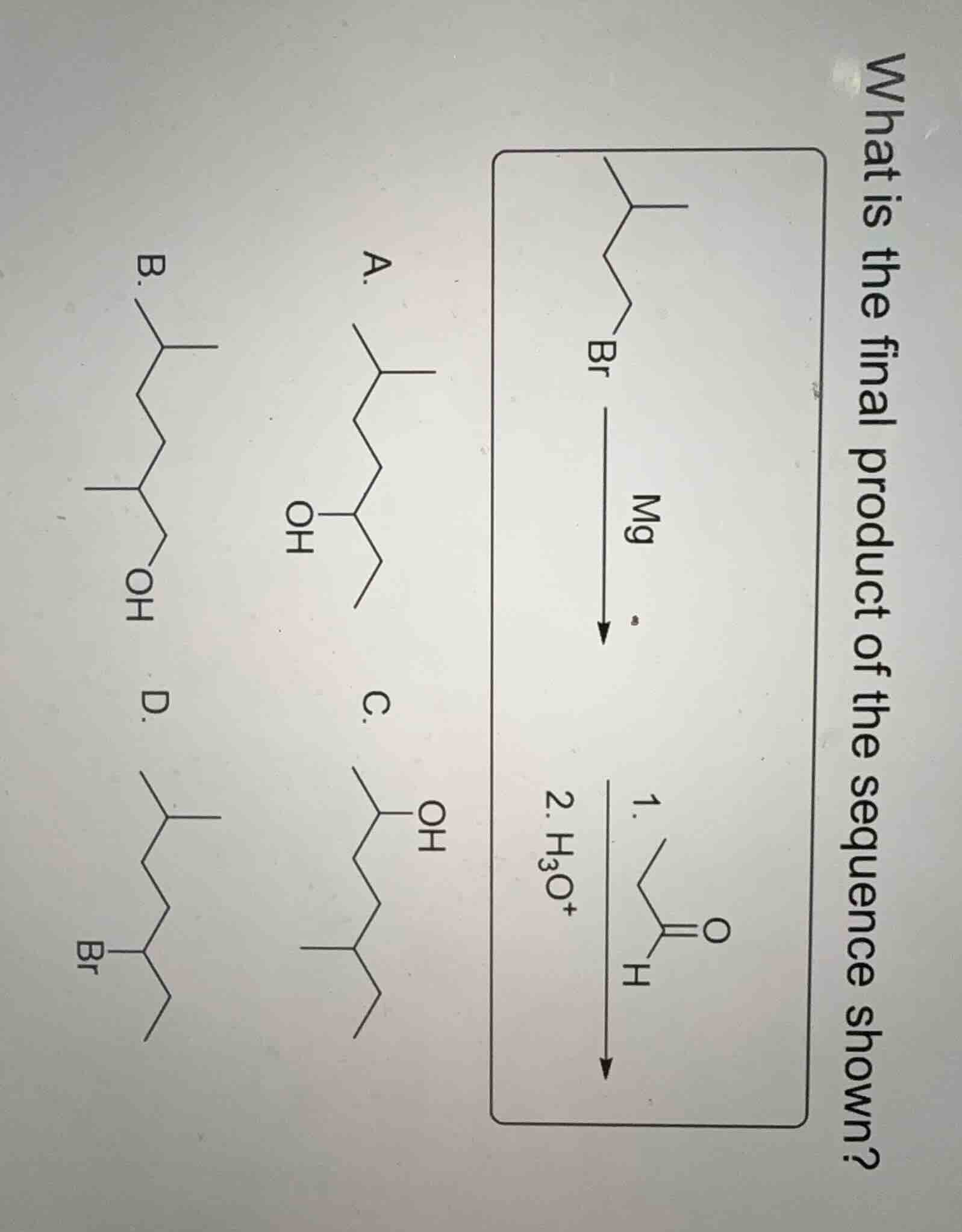
Question
what is the final product of the sequence shown?
chemical reaction and structure diagrams omitted
a. structure a
b. structure b
c. structure c
d. structure d
Step1: Form Grignard Reagent
The alkyl bromide reacts with Mg to form a Grignard reagent. The reaction is \( \text{R-Br} + \text{Mg}
ightarrow \text{R-MgBr} \), where R is the alkyl group from the bromide.
Step2: React with Aldehyde
The Grignard reagent (a nucleophile) attacks the carbonyl carbon of the aldehyde (\( \text{CH}_3\text{CH}_2\text{CHO} \)). This forms an alkoxide intermediate after the addition.
Step3: Protonation with \( \text{H}_3\text{O}^+ \)
The alkoxide intermediate is protonated by \( \text{H}_3\text{O}^+ \) to form a secondary alcohol. By analyzing the structures, option A has the correct carbon skeleton and the -OH group at the appropriate position (resulting from the addition to the aldehyde and subsequent protonation).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. The structure with the alkyl chain and -OH group as shown in option A (matching the product of Grignard addition to aldehyde followed by protonation).